Cytokinetics Presents Additional Data From GALACTIC-HF at the American Heart Association Scientific Sessions 2024
Rhea-AI Summary
Cytokinetics (CYTK) presented new post-hoc analyses of GALACTIC-HF, their Phase 3 trial of omecamtiv mecarbil for heart failure, at AHA Scientific Sessions 2024. The analyses showed that the drug reduced risk of adverse cardiac outcomes in severe heart failure patients regardless of age. Among 8,232 patients studied over 21.8 months, the treatment demonstrated significant risk reductions in both age groups (<65 and ≥65 years). The drug also showed potential benefits in reducing ventricular arrhythmias (VA) risk in patients with severely reduced left ventricular ejection fraction, while maintaining a favorable safety profile.
Positive
- Treatment showed significant risk reduction in primary outcomes for both age groups (HR 0.77 for <65 years, HR 0.83 for ≥65 years)
- Large absolute risk reductions observed (8.6 and 7.9 respectively) in severe heart failure patients
- Drug demonstrated potential to reduce risk of ventricular arrhythmias in patients with severely reduced LVEF
- No increase in adverse events compared to placebo across age groups
Negative
- None.
News Market Reaction – CYTK
On the day this news was published, CYTK gained 3.42%, reflecting a moderate positive market reaction.
Data tracked by StockTitan Argus on the day of publication.
SOUTH SAN FRANCISCO, Calif., Nov. 16, 2024 (GLOBE NEWSWIRE) -- Cytokinetics, Incorporated (Nasdaq: CYTK) today announced new data from post-hoc analyses of GALACTIC-HF (Global Approach to Lowering Adverse Cardiac Outcomes Through Improving Contractility in Heart Failure), the Phase 3 cardiovascular outcomes clinical trial of omecamtiv mecarbil were presented at the American Heart Association Scientific Sessions 2024 in Chicago, IL.
“These analyses reinforce the potential treatment benefit of omecamtiv mecarbil in patients from GALACTIC-HF who are at higher risk, such as older patients or those with a recent ventricular arrhythmia,” said Stuart Kupfer, M.D., Senior Vice President, Chief Medical Officer. “As we prepare for the start of COMET-HF, the confirmatory Phase 3 clinical trial of omecamtiv mecarbil, we look forward to evaluating its potential to reduce the risk of adverse heart failure outcomes across the spectrum of these very high-risk patients failing guideline-directed medical therapy.”
Treatment with Omecamtiv Mecarbil Reduced Risk of the Primary Composite Endpoint in Patients with Severe Heart Failure Independent of Age
A post-hoc analysis from GALACTIC-HF supports the potential efficacy of omecamtiv mecarbil irrespective of age, including in patients with severe heart failure. Of the 8,232 patients included in the analysis, patients were on average 64.5 years old, with
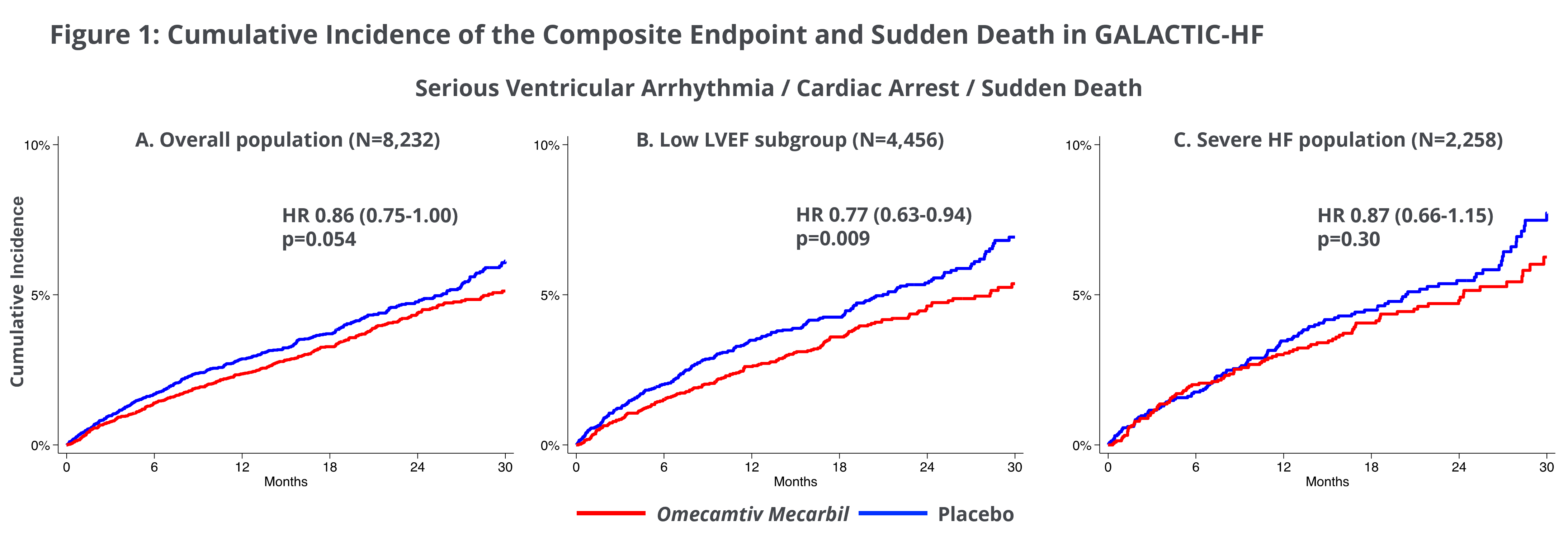
Ventricular Arrythmias Associated with Adverse Clinical Outcomes in Patients with Heart Failure and Reduced Ejection Fraction; Treatment with Omecamtiv Mecarbil Reduces Risk of Ventricular Arrythmias in Patients with Severely Reduced LVEF
A second post-hoc analysis from GALACTIC-HF investigated clinical consequences of ventricular arrhythmias (VA) in the patients studied in GALACTIC-HF. Of the 8,232 patients enrolled in GALACTIC-HF, following an occurrence of VA, there was a significantly higher risk of the primary endpoint of cardiovascular death or heart failure events (HR 1.67;
About Omecamtiv Mecarbil
Omecamtiv mecarbil is an investigational, selective, small molecule cardiac myosin activator, the first of a novel class of myotropes1 designed to directly target the contractile mechanisms of the heart, binding to and recruiting more cardiac myosin heads to interact with actin during systole. Omecamtiv mecarbil is designed to increase the number of active actin-myosin cross bridges during each cardiac cycle and consequently augment the impaired contractility that is associated with heart failure with reduced ejection fraction (HFrEF). Preclinical research has shown that omecamtiv mecarbil increases cardiac contractility without increasing intracellular myocyte calcium concentrations or myocardial oxygen consumption.2-4
The development program for omecamtiv mecarbil assessed its potential for the treatment of HFrEF. Positive results from GALACTIC-HF, the first Phase 3 clinical trial of omecamtiv mecarbil demonstrated a statistically significant effect of treatment with omecamtiv mecarbil to reduce risk of the primary composite endpoint of cardiovascular (CV) death or heart failure events (heart failure hospitalization and other urgent treatment for heart failure) compared to placebo in patients treated with standard of care. No reduction in the secondary endpoint of time to CV death was observed. In general, the overall rates of myocardial ischemia, ventricular arrhythmias and death were similar between treatment and placebo groups. Adverse events and treatment discontinuation of study drug were balanced between treatment arms. The magnitude of the treatment effect in a pre-specified subgroup of more than 4,000 patients with heart failure with severely reduced ejection fraction (<
About Heart Failure with Severely Reduced Ejection Fraction
Heart failure is a grievous condition that affects more than 64 million people worldwide5 about half of whom have reduced left ventricular function.6,7 It is the leading cause of hospitalization and readmission in people age 65 and older.8,9 By 2029 is it estimated that 2.8 million people in the U.S. will have heart failure with severely reduced ejection fraction10, characterized as heart failure with reduced ejection fraction (HFrEF) <
About Cytokinetics
Cytokinetics is a late-stage, specialty cardiovascular biopharmaceutical company focused on discovering, developing and commercializing muscle biology-directed drug candidates as potential treatments for debilitating diseases in which cardiac muscle performance is compromised. As a leader in muscle biology and the mechanics of muscle performance, the company is developing small molecule drug candidates specifically engineered to impact myocardial muscle function and contractility. Following positive results from SEQUOIA-HCM, the pivotal Phase 3 clinical trial evaluating aficamten, a next-in-class cardiac myosin inhibitor, in obstructive hypertrophic cardiomyopathy (HCM), Cytokinetics submitted an NDA for aficamten to the U.S. Food & Drug Administration and is progressing regulatory submissions for aficamten for the treatment of obstructive HCM in Europe. Aficamten is also currently being evaluated in MAPLE-HCM, a Phase 3 clinical trial of aficamten as monotherapy compared to metoprolol as monotherapy in patients with obstructive HCM, ACACIA-HCM, a Phase 3 clinical trial of aficamten in patients with non-obstructive HCM, CEDAR-HCM, a clinical trial of aficamten in a pediatric population with obstructive HCM, and FOREST-HCM, an open-label extension clinical study of aficamten in patients with HCM. Cytokinetics is also developing omecamtiv mecarbil, a cardiac muscle activator, in patients with heart failure with severely reduced ejection fraction (HFrEF), CK-586, a cardiac myosin inhibitor with a mechanism of action distinct from aficamten for the potential treatment of heart failure with preserved ejection fraction (HFpEF), and CK-089, a fast skeletal muscle troponin activator (FSTA) for the potential treatment of a specific type of muscular dystrophy.
For additional information about Cytokinetics, visit www.cytokinetics.com and follow us on X, LinkedIn, Facebook and YouTube.
Forward-Looking Statements
This press release contains forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995 (the "Act"). Cytokinetics disclaims any intent or obligation to update these forward-looking statements and claims the protection of the Act's Safe Harbor for forward-looking statements. Examples of such statements include, but are not limited to, statements, express or implied, relating to the Company’s development plans for omecamtiv mecarbil in the United States. Such statements are based on management's current expectations, but actual results may differ materially due to various risks and uncertainties, including, but not limited to, potential difficulties or delays in the development, testing, regulatory approvals for trial commencement, progression or product sale or manufacturing, or production of Cytokinetics' drug candidates that could slow or prevent clinical development or product approval; Cytokinetics' drug candidates may have adverse side effects or inadequate therapeutic efficacy; the FDA or foreign regulatory agencies may delay or limit Cytokinetics' ability to conduct clinical trials; Cytokinetics may be unable to obtain or maintain patent or trade secret protection for its intellectual property; standards of care may change, rendering Cytokinetics' drug candidates obsolete; and competitive products or alternative therapies may be developed by others for the treatment of indications Cytokinetics' drug candidates and potential drug candidates may target. For further information regarding these and other risks related to Cytokinetics' business, investors should consult Cytokinetics' filings with the Securities and Exchange Commission.
CYTOKINETICS® and the C-shaped logo are registered trademarks of Cytokinetics in the U.S. and certain other countries.
Contact:
Cytokinetics
Diane Weiser
Senior Vice President, Corporate Affairs
(415) 290-7757
References:
- Psotka MA, Gottlieb SS, Francis GS et al. Cardiac Calcitropes, Myotropes, and Mitotropes. JACC. 2019; 73:2345-53.
- Planelles-Herrero VJ, Hartman JJ, Robert-Paganin J. et al. Mechanistic and structural basis for activation of cardiac myosin force production by omecamtiv mecarbil. Nat Commun. 2017;8:190.
- Shen YT, Malik FI, Zhao X, et al. Improvement of cardiac function by a cardiac myosin activator in conscious dogs with systolic heart failure. Circ Heart Fail. 2010; 3: 522-27.
- Malik FI, Hartman JJ, Elias KA, Morgan BP, Rodriguez H, Brejc K, Anderson RL, Sueoka SH, Lee KH, Finer JT, Sakowicz R. Cardiac myosin activation: a potential therapeutic approach for systolic heart failure. Science. 2011 Mar 18;331(6023):1439-43.
- James et al. GBD 2017 Disease and Injury Incidence and Prevalence Collaborators. Lancet 2018; 392: 1789–858.
- Yancy CW, Jessup M, Bozkurt B, et al. 2013 ACCF/AHA Guideline for the Management of Heart failure: A Report of the American College of Cardiology Foundation/American Heart Association Task Force on Practice Guidelines. Circulation. 2013;128:e240-e327.
- Ponikowski P, Voors AA, Anker SD, et al. 2016 ESC guidelines for the diagnosis and treatment of acute and chronic heart failure: The Task Force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC). Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J. 2016;37:2129–2200.
- Roger VL. Epidemiology of Heart Failure. Circulation Research. 2013;113:646-659, originally published August 29, 2013. doi: 10.1161/CIRCRESAHA.113.300268.
- Kilgore M, Patel HK, Kielhorn A, et al. Economic burden of hospitalizations of Medicare beneficiaries with heart failure. Risk Manag Healthc Policy. 2017; 10: 63-70.
- Taylor C J, Ordóñez-Mena J M, Roalfe A K, et al. Trends in survival after a diagnosis of heart failure in the United Kingdom 2000-2017: population based cohort study. BMJ 2019; 364:l223 doi:10.1136/bmj.l223
- Greene SJ, Bauersachs J, Brugts JJ, et al. Worsening Heart Failure: Nomenclature, Epidemiology, and Future Directions: JACC Review Topic of the Week. JACC. 2023 Jan 31;81(4):413-424. doi:10.1016/j.jacc.2022.11.023. PMID: 36697141.
- Extrapolated from Desai NR, Butler J, Binder G, et al. Prevalence and Excess Risk of Hospitalization in Heart Failure with Reduced Ejection Fraction. Poster presented at: Heart Failure Society of America (HFSA) Annual Scientific Meeting; 2022 Sep 30-Oct 3; Washington, DC.
- Carnicelli AP, Clare RM, Hofmann P, et al. Clinical trajectory of patients with a worsening heart failure event and reduced ventricular ejection fraction. Am Heart J. 2022 Mar; 245:110-116. doi: 10.1016/j.ahj.2021.12.003. Epub 2021 Dec 18. PMID: 34932997.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/ee1006b4-fa47-4800-8929-5cdb837d546e

FAQ
What were the key findings of CYTK's GALACTIC-HF trial presented at AHA 2024?
How many patients were included in CYTK's GALACTIC-HF analysis?
What was the median follow-up period in CYTK's GALACTIC-HF trial?







