Autonomix Secures Principal Investigators and Completes Clinical Training for First Human Study
Proof-of-concept (PoC) human clinical trial targeting multi-billion-dollar pancreatic cancer market through transvascular RF ablation
Company expects to commence patient enrollment imminently
THE WOODLANDS, TX, Feb. 22, 2024 (GLOBE NEWSWIRE) -- Autonomix Medical, Inc. (NASDAQ: AMIX) (“Autonomix” or the “Company”), a medical device company focused on advancing innovative technologies to revolutionize how diseases involving the nervous system are diagnosed and treated, today announced it has completed selection of Principal Investigators for its proof-of-concept human clinical study evaluating the use of transvascular RF ablation for the treatment of pancreatic cancer pain. Additionally, the Principal Investigators have successfully completed training for the PoC study.
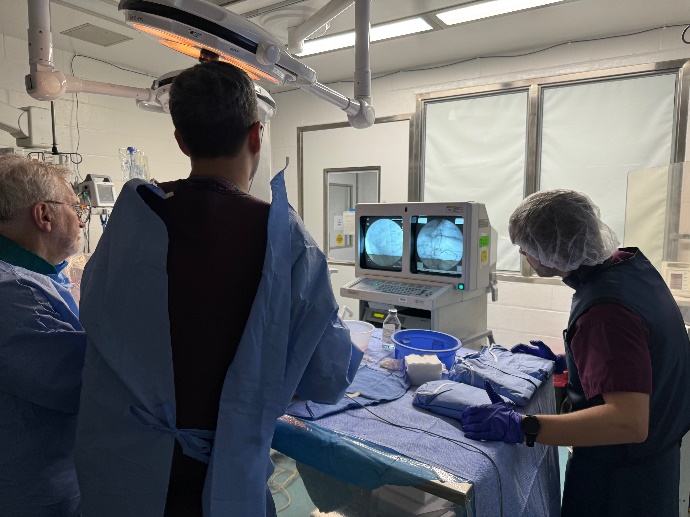
“We are building important momentum with the advancement of our PoC human clinical study. We worked diligently to identify and secure our Principal Investigators for the study and have completed the training for this first study ever to evaluate RF ablation in a transvascular approach. Our lead investigator performing the procedure is not only trained in interventional radiology, but he is also a cardiac surgeon. With these critical steps now completed, we remain on track to commence patient enrollment this quarter,” commented Lori Bisson, Chief Executive Officer of Autonomix.
The primary objective of the proof-of-concept human clinical study is to successfully ablate relevant somatic nerves and mitigate pain in patients with pancreatic cancer pain utilizing RF ablation in a transvascular approach to the nerves in the region. Twenty (20) subjects will be enrolled at one clinical trial site for the study. Confirmation of suitability will be affirmed by the primary oncology service caring for the patients. Up to 5 additional patients will be included and treated according to protocol to ensure the physician’s familiarity with the procedure. However, they will not be included in the analysis of the study objectives. Enrollment is expected to be completed before the end of 2024.
Expected Upcoming Milestones
- Q1 CY2024: Commence enrollment in PoC human clinical study evaluating proprietary ablation technology for treatment of pancreatic cancer pain
- 2024: Complete ablation device design intended for clinical use
- 2025: Complete development of ablation system and catheter in preparation for human pivotal trial
- 2025: Launch combined sensing / ablation clinical trial
- 2026: De novo submission
- 2027: Seek FDA clearance
The Company’s catheter-based technology is being developed to do two things: sense neural signals associated with pain or disease and precisely target those nerves for treatment. Autonomix believes this technology is a better alternative to the current approaches commonly used today, where doctors either rely on systemic drugs like opioids that lose effectiveness and have unwanted side effects or treat suspected areas blindly in hopes of hitting the right nerves, an approach that is often inaccurate and can miss the target and even cause collateral damage to surrounding parts of the body.
The Company is initially developing its technology to address pancreatic cancer-related pain. Current approaches, primarily relying on opioids or invasive ethanol injections, can provide only limited relief and may lead to risky side effects. For more information about the Company’s technology, please visit autonomix.com.
About Autonomix Medical, Inc.
Autonomix is a medical device company focused on advancing innovative technologies to revolutionize how diseases involving the nervous system are diagnosed and treated. The Company’s first-in-class technology platform includes a catheter-based microchip sensing array that has the ability to detect and differentiate neural signals with approximately 3,000 times greater sensitivity than currently available technologies. We believe this will enable, for the first time ever, transvascular diagnosis and treatment of diseases involving the peripheral nervous system virtually anywhere in the body.
We are initially developing our technology for pancreatic cancer pain, a condition that can cause debilitating pain and needs an effective solution. However, our technology constitutes a platform with the potential to address dozens of indications, including in cardiology, renal denervation and chronic pain management across a wide disease spectrum.
For more information, visit autonomix.com and connect with the Company on X, LinkedIn, Instagram and Facebook.
Forward Looking Statements
Some of the statements in this release are “forward-looking statements,” which involve risks and uncertainties. Forward-looking statements in this press release include, without limitation, the timing of the completion of patient enrollment in the study and the Company’s ability to successfully meet the milestones set forth in this press release on a timely basis, if at all. Such forward-looking statements can be identified by the use of words such as ‘should,’ ‘may,’ ‘intends,’ ‘anticipates,’ ‘believes,’ ‘estimates,’ ‘projects,’ ‘forecasts,’ ‘expects,’ ‘plans,’ and ‘proposes.’
Although Autonomix Medical, Inc. (or Autonomix) believes that the expectations reflected in these forward-looking statements are based on reasonable assumptions, there are a number of risks and uncertainties that could cause actual results to differ materially from such forward-looking statements. You are urged to carefully review and consider any cautionary statements and other disclosures, including the statements made under the heading “Risk Factors” and elsewhere in the offering statement filed with the U.S. Securities and Exchange Commission (“SEC”). Forward-looking statements speak only as of the date of the document in which they are contained and Autonomix does not undertake any duty to update any forward-looking statements except as may be required by law.
Investor and Media Contact
JTC Team, LLC
Jenene Thomas
833-475-8247
autonomix@jtcir.com
Attachment

