ExoPTEN Preclinical Study Demonstrates Significant Potential for Enhancing Motor Function, Blood Flow, and Spinal Cord Injury Recovery
NurExone Biologic Inc. (NRXBF) has completed a important preclinical study for its ExoPTEN treatment, marking progress toward IND submission and first-in-human trials. The study demonstrated that ExoPTEN significantly improved both motor function recovery and blood flow at spinal cord injury sites.
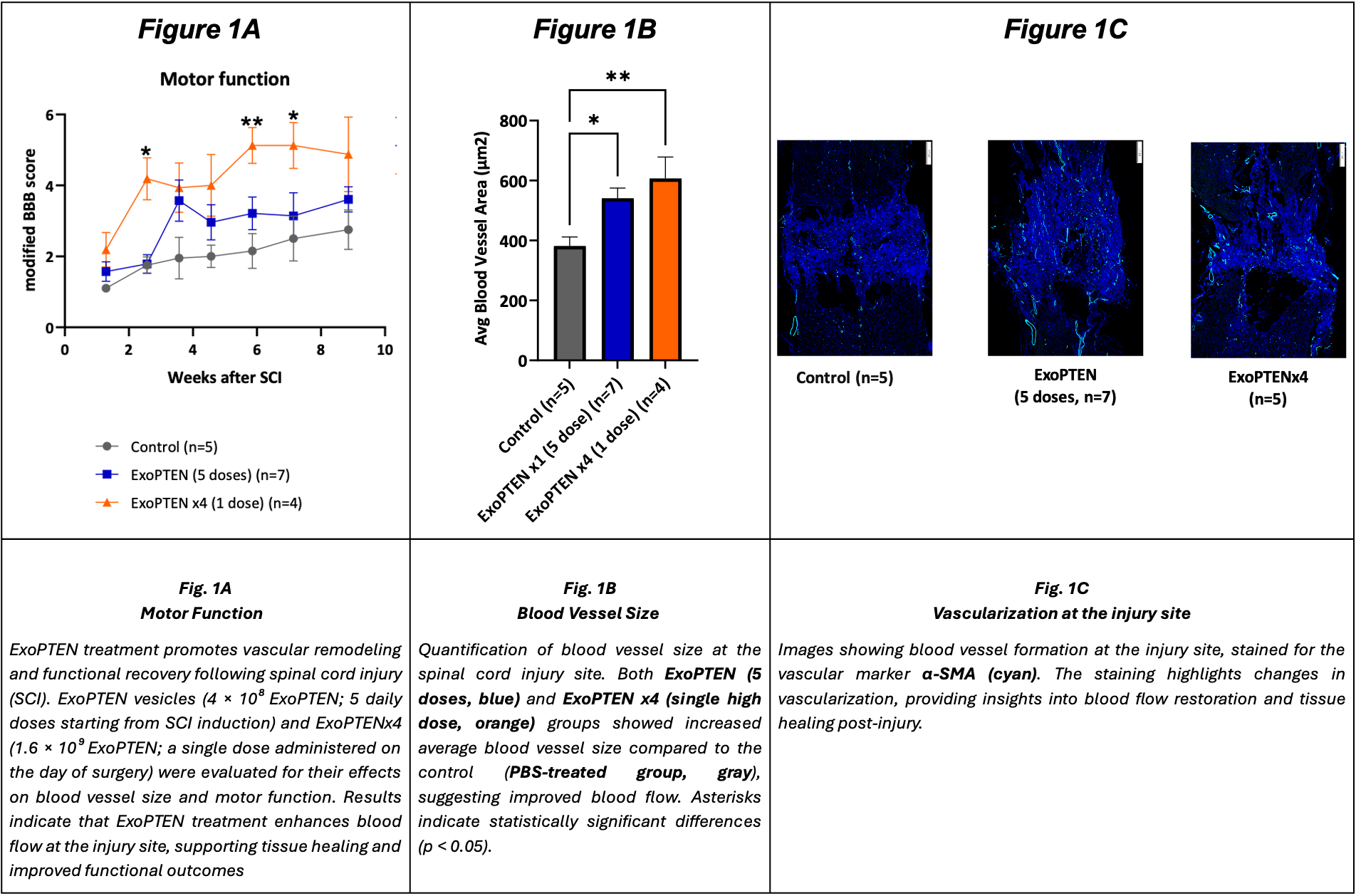
The research compared two dosing regimens: a single high dose on surgery day versus a lower dose over five consecutive days. Both groups showed notable improvements in motor function recovery compared to the control group, measured by the modified BBB locomotor rating scale. Histological analysis revealed increased average blood vessel size in treated subjects, indicating enhanced circulation important for tissue healing.
Dr. Tali Kizhner, Director of R&D at NurExone, highlighted that the results support ExoPTEN's potential to enhance natural repair mechanisms following spinal cord injury. The company will continue refining ExoPTEN's therapeutic profile as part of its ongoing preclinical program.
NurExone Biologic Inc. (NRXBF) ha completato un importante studio preclinico per il suo trattamento ExoPTEN, segnando un progresso verso la presentazione dell'IND e i primi trial sull'uomo. Lo studio ha dimostrato che ExoPTEN ha migliorato significativamente sia il recupero della funzione motoria che il flusso sanguigno nei siti di lesione del midollo spinale.
La ricerca ha confrontato due regimi di dosaggio: una singola dose elevata il giorno dell'intervento rispetto a una dose più bassa distribuita su cinque giorni consecutivi. Entrambi i gruppi hanno mostrato miglioramenti notevoli nel recupero della funzione motoria rispetto al gruppo di controllo, misurati attraverso la scala di valutazione locomotoria BBB modificata. L'analisi istologica ha rivelato un aumento della dimensione media dei vasi sanguigni nei soggetti trattati, indicando una circolazione migliorata importante per la guarigione dei tessuti.
Il Dr. Tali Kizhner, Direttore R&D di NurExone, ha sottolineato che i risultati supportano il potenziale di ExoPTEN di migliorare i meccanismi di riparazione naturale dopo una lesione del midollo spinale. L'azienda continuerà a perfezionare il profilo terapeutico di ExoPTEN come parte del suo programma preclinico in corso.
NurExone Biologic Inc. (NRXBF) ha completado un importante estudio preclínico para su tratamiento ExoPTEN, marcando un avance hacia la presentación del IND y los primeros ensayos en humanos. El estudio demostró que ExoPTEN mejoró significativamente tanto la recuperación de la función motora como el flujo sanguíneo en los sitios de lesión de la médula espinal.
La investigación comparó dos regímenes de dosificación: una sola dosis alta el día de la cirugía frente a una dosis más baja durante cinco días consecutivos. Ambos grupos mostraron mejoras notables en la recuperación de la función motora en comparación con el grupo de control, medida a través de la escala de calificación locomotora BBB modificada. El análisis histológico reveló un aumento en el tamaño promedio de los vasos sanguíneos en los sujetos tratados, lo que indica una circulación mejorada importante para la curación de los tejidos.
El Dr. Tali Kizhner, Director de I+D en NurExone, destacó que los resultados respaldan el potencial de ExoPTEN para mejorar los mecanismos de reparación natural tras una lesión de la médula espinal. La empresa continuará perfeccionando el perfil terapéutico de ExoPTEN como parte de su programa preclínico en curso.
NurExone Biologic Inc. (NRXBF)는 ExoPTEN 치료를 위한 중요한 전임상 연구를 완료하였으며, 이는 IND 제출 및 인간 대상 첫 번째 임상 시험을 향한 진전을 의미합니다. 연구 결과 ExoPTEN이 척수 손상 부위에서 운동 기능 회복과 혈류를 모두 유의미하게 개선했다는 사실이 입증되었습니다.
연구에서는 두 가지 투여 요법을 비교했습니다: 수술 당일에 단일 고용량을 투여하는 것과 5일 연속으로 낮은 용량을 투여하는 것입니다. 두 그룹 모두 대조군에 비해 수정된 BBB 보행 평가 척도로 측정된 운동 기능 회복에서 주목할 만한 개선을 보였습니다. 조직학적 분석 결과, 치료받은 피험자에서 평균 혈관 크기가 증가하여 조직 치유에 중요한 개선된 순환을 나타냈습니다.
NurExone의 R&D 책임자인 Tali Kizhner 박사는 결과가 척수 손상 후 자연 치유 메커니즘을 향상시킬 수 있는 ExoPTEN의 잠재력을 뒷받침한다고 강조했습니다. 회사는 진행 중인 전임상 프로그램의 일환으로 ExoPTEN의 치료 프로필을 계속해서 다듬어 나갈 것입니다.
NurExone Biologic Inc. (NRXBF) a complété une étude préclinique importante pour son traitement ExoPTEN, marquant un progrès vers la soumission de l'IND et les premiers essais sur l'homme. L'étude a démontré qu'ExoPTEN améliorait de manière significative à la fois la récupération de la fonction motrice et le flux sanguin sur les sites de lésions de la moelle épinière.
La recherche a comparé deux schémas posologiques : une seule forte dose le jour de la chirurgie par rapport à une dose plus faible sur cinq jours consécutifs. Les deux groupes ont montré des améliorations notables dans la récupération de la fonction motrice par rapport au groupe de contrôle, mesurées à l'aide de l'échelle de notation locomotrice BBB modifiée. L'analyse histologique a révélé une augmentation de la taille moyenne des vaisseaux sanguins chez les sujets traités, indiquant une circulation améliorée importante pour la guérison des tissus.
Le Dr Tali Kizhner, Directeur de la R&D chez NurExone, a souligné que les résultats soutiennent le potentiel d'ExoPTEN à améliorer les mécanismes de réparation naturels après une lésion de la moelle épinière. L'entreprise continuera à affiner le profil thérapeutique d'ExoPTEN dans le cadre de son programme préclinique en cours.
NurExone Biologic Inc. (NRXBF) hat eine wichtige präklinische Studie für seine ExoPTEN-Behandlung abgeschlossen, was einen Fortschritt in Richtung IND-Einreichung und erster klinischer Versuche am Menschen darstellt. Die Studie zeigte, dass ExoPTEN sowohl die motorische Funktionswiederherstellung als auch den Blutfluss an den Stellen von Rückenmarksverletzungen signifikant verbesserte.
Die Forschung verglich zwei Dosierungsregime: eine einzelne hohe Dosis am Operationstag im Vergleich zu einer niedrigeren Dosis über fünf aufeinanderfolgende Tage. Beide Gruppen zeigten im Vergleich zur Kontrollgruppe bemerkenswerte Verbesserungen bei der motorischen Funktionswiederherstellung, gemessen an der modifizierten BBB-Lokomotorbewertungsskala. Histologische Analysen zeigten eine erhöhte durchschnittliche Größe der Blutgefäße bei behandelten Probanden, was auf eine verbesserte Durchblutung hinweist, die für die Gewebeheilung wichtig ist.
Dr. Tali Kizhner, Direktor für Forschung und Entwicklung bei NurExone, betonte, dass die Ergebnisse das Potenzial von ExoPTEN unterstützen, die natürlichen Reparaturmechanismen nach einer Rückenmarksverletzung zu verbessern. Das Unternehmen wird das therapeutische Profil von ExoPTEN im Rahmen seines laufenden präklinischen Programms weiter verfeinern.
- Successful completion of important preclinical study for IND submission
- Both dosing regimens showed significant motor function improvements
- Demonstrated increased blood vessel size and improved circulation
- Progress toward first-in-human trials
- Still in preclinical phase, requiring further studies before human trials
- Multiple regulatory approvals still needed before commercialization
TORONTO and HAIFA, Israel, March 14, 2025 (GLOBE NEWSWIRE) -- NurExone Biologic Inc. (TSXV: NRX) (OTCQB: NRXBF) (FSE: J90) (“NurExone” or the “Company”) is pleased to announce that it has successfully completed an important preclinical study towards its Investigational New Drug (“IND”) submission. The new study, which advances the Company’s path towards first-in-human trials, demonstrated that ExoPTEN treatment with different dose regimens led to both motor function recovery and significant improvements in blood flow at the site of spinal cord injury—an essential factor in tissue healing and functional recovery.i
“This preclinical study evaluated dosing regimens to provide efficacy data in support of our IND submission,” said Dr. Tali Kizhner, Director of R&D at NurExone. “The results reinforce ExoPTEN’s potential to enhance the body’s natural repair mechanisms following spinal cord injury. Notably, the increased blood vessel size observed in treated subjects indicated improved circulation, which is crucial for oxygen and nutrient delivery to damaged tissues. These findings suggest that ExoPTEN has the potential to become a transformative therapeutic candidate, and we are eager to advance toward clinical trials.”
Scientific publications and reach in the field have shown already that post-injury angiogenesis and vascular remodeling correlate with improved functional recovery in spinal cord injury models.ii
The study compared two dosing regimens of ExoPTEN: a single high dose on the day of surgery versus a lower dose administered over five consecutive days. Both treatment groups showed significant improvements in motor function recovery compared to the control group, as measured by the modified Basso, Beattie, and Bresnahan (“BBB”) locomotor rating scale (Figure 1A). Additionally, histological analysis revealed that ExoPTEN treatment significantly increased the average blood vessel size (Figure 1B-1C), suggesting improved circulationi - a critical factor in post-injury healing and functional restoration.
NurExone will continue to refine ExoPTEN’s therapeutic profile as part of its ongoing preclinical program, paving the way to IND submission and regulatory approval for first-in-human trials.
About NurExone
NurExone Biologic Inc. is a TSX Venture Exchange (“TSXV”), OTCQB and Frankfurt-listed biotech company focused on developing regenerative exosome-based therapies for central nervous system injuries. Its lead product, ExoPTEN, has demonstrated strong preclinical data supporting clinical potential in treating acute spinal cord and optic nerve injury, both multi-billion-dollar marketsiii. Regulatory milestones, including Orphan Drug Designation, facilitate the roadmap towards clinical trials in the U.S. and Europe. Commercially, the Company is expected to offer solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications. NurExone has established Exo-Top Inc., a U.S. subsidiary, to anchor its North American activity and growth strategy.
For additional information and a brief interview, please watch Who is NurExone?, visit www.nurexone.com or follow NurExone on LinkedIn, Twitter, Facebook, or YouTube.
For more information, please contact:
Dr. Lior Shaltiel
Chief Executive Officer and Director
Phone: +972-52-4803034
Email: info@nurexone.com
Oak Hill Financial Inc.
2 Bloor Street, Suite 2900
Toronto, Ontario M4W 3E2
Investor Relations – Canada
Phone: +1-647-479-5803
Email: info@oakhillfinancial.ca
Dr. Eva Reuter
Investor Relations – Germany
Phone: +49-69-1532-5857
Email: e.reuter@dr-reuter.eu
Allele Capital Partners
Investor Relations – U.S.
Phone: +1 978-857-5075
Email: aeriksen@allelecapital.com
FORWARD-LOOKING STATEMENTS
This press release contains certain “forward-looking statements” that reflect the Company’s current expectations and projections about its future results. Wherever possible, words such as “may”, “will”, “should”, “could”, “expect”, “plan”, “intend”, “anticipate”, “believe”, “estimate”, “predict” or “potential” or the negative or other variations of these words, or similar words or phrases, have been used to identify these forward-looking statements. Forward-looking statements in this press release include, but are not limited to, statements relating to: the results of the Company’s preclinical trials; the Company completing an IND submission; the Company advancing towards clinical trials and launching a first-in-human trial; the anticipated benefits of ExoPTEN; the Company continuing to refine its product candidates; the Company receiving requisite regulatory approvals; the Company’s future plans and expectations; NurExone’s focus on developing regenerative exosome-based therapies for central nervous system injuries; and the NurExone platform technology offering solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications.
These statements reflect management’s current beliefs and are based on information currently available to management as at the date hereof. In developing the forward-looking statements in this press release, we have applied several material assumptions, including: the Company will realize upon the stated benefits of the preclinical trials and such clinical trials will have the intended results; the Company will complete an IND submission; the Company will advance towards clinical trials and launch a first-in-human trial; ExoPTEN will have its anticipated benefits; the Company will continue to refine its product candidates; the Company will receive requisite regulatory approvals; the Company will continue to succeed; NurExone will continue to focus on developing regenerative exosome-based therapies for central nervous system injuries; and the NurExone platform technology will offer solutions to companies interested in quality exosomes and minimally invasive targeted delivery systems for other indications.
Forward-looking statements involve significant risk, uncertainties and assumptions. Many factors could cause actual results, performance or achievements to differ materially from the results discussed or implied in the forward-looking statements. These risks and uncertainties include, but are not limited to risks related to: the Company’s early stage of development; lack of revenues to date; government regulation; market acceptance for its products; rapid technological change; dependence on key personnel, and on the Company’s strategic partners; the fact that preclinical drug development is uncertain, and the drug product candidates of the Company may never advance to clinical trials or human trials; the fact that results of preclinical studies and early-stage clinical trials may not be predictive of the results of later stage clinical trials; the uncertain outcome, cost, and timing of product development activities, preclinical studies and clinical trials of the Company; the uncertain clinical development process, including the risk that clinical trials may not have an effective design or generate positive results; the inability to obtain or maintain regulatory approval of the drug product candidates of the Company; the introduction of competing drugs that are safer, more effective or less expensive than, or otherwise superior to, the drug product candidates of the Company; the initiation, conduct, and completion of preclinical studies and clinical trials may be delayed, adversely affected or impacted by unforeseen issues; the inability to obtain or maintain intellectual property protection for the drug product candidates of the Company; risks that the Company’s intellectual property and technology won’t have the intended impact on the Company and/or its business; the Company’s inability to carry out its preclinical trials and/or realize upon the stated benefits of the preclinical trials and/or such preclinical trials will not have the intended results; the inability of the Company to fulfill its intended future plans and expectations; the Company will be unable to complete an IND submission; ExoPTEN will not have its anticipated benefits; the Company will be unable to refine its product candidates; NurExone being unable to focus on developing regenerative exosome-based therapies for central nervous system injuries; the NurExone platform technology being unable to offer solutions to companies interested in quality exosomes and/or minimally invasive targeted delivery systems for other indications; and the risks discussed under the heading “Risk Factors” on pages 44 to 51 of the Company’s annual information form dated August 27, 2024, a copy of which is available under the Company’s SEDAR+ profile at www.sedarplus.ca. These factors should be considered carefully, and readers should not place undue reliance on the forward-looking statements. Although the forward-looking statements contained in this press release are based upon what management believes to be reasonable assumptions, the Company cannot assure readers that actual results will be consistent with these forward-looking statements. These forward-looking statements are made as of the date of this press release, and the Company assumes no obligation to update or revise them to reflect new events or circumstances, except as required by law.
Neither TSXV nor its Regulation Services Provider (as that term is defined in the policies of the TSXV) accepts responsibility for the adequacy or accuracy of this release.
_______________________
i Blood-spinal cord barrier after spinal cord injury: relation to revascularization and wound healing, J Neurosci Res, 2003, https://pubmed.ncbi.nlm.nih.gov/14515352/
ii Blood-Spinal Cord Barrier in Spinal Cord Injury: A Review, J Neurotrauma, 2021, https://pubmed.ncbi.nlm.nih.gov/33292072/
iii Spinal cord injury, Glaucoma
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/72e6aea2-8b07-47ab-a582-fbc21aa63c72








