Orthofix Announces 510k Clearance and Full Commercial Launch of OsteoCove, an Advanced Bioactive Synthetic Graft for use in Spine and Orthopedic Procedures
- OsteoCove is formulated to provide superior bone-forming capabilities and has been cleared for commercial use.
- The product features a specialized granule surface topography designed to promote bone formation.
- Orthofix expects strong growth in the $300 million U.S. synthetic bone graft market with the introduction of OsteoCove.
- None.
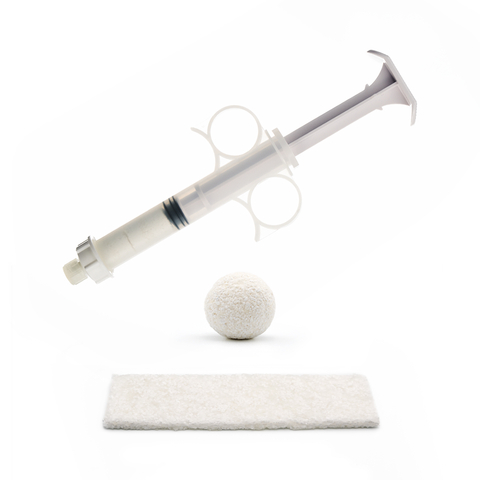
Image of OsteoCove putty and strip, an advanced bioactive synthetic graft formulated to provide superior bone-forming capabilities with best-in-class handling characteristics for a wide range of spine and orthopedic procedural applications. (Photo: Business Wire)
“Using the same disciplined R&D approach we have historically taken with our internally processed demineralized bone matrix solutions, we created a product that challenges the performance expectations of typical synthetic bone graft substitutes,” said Dr. Frank Vizesi, Chief Scientific Officer for Orthofix. “OsteoCove is another prime example of leveraging our strengths in product development in combination with our vertically integrated manufacturing expertise.”
Made of biphasic ceramic granule comprised of β-tricalcium phosphate (ß-TCP) and hydroxyapatite (HA) combined with type-I bovine collagen, OsteoCove features a specialized granule surface topography designed to elicit a bone-forming response, as evidenced by its ability to grow bone in a challenging muscle pouch model. This specialized surface chemistry and microporosity are shown to promote superior bone formation when compared to other commercially available advanced synthetic grafts.1
“Orthofix is a leader in both the demineralized bone matrices (DBM) and cellular allograft markets, and the launch of OsteoCove significantly strengthens our biologics portfolio and builds on our mission to deliver best-in-class products in every major bone grafting category,” said Tyler Lipschultz, President, Orthofix Global Biologics. “With the introduction of OsteoCove, we expect strong growth in this large market segment as we continue to deliver on our commitment to provide surgeons a comprehensive offering of biologic solutions to meet the needs of their patients.”
The
Orthofix invites those attending the North American Spine Surgeon Annual Meeting in
1,2 Data on file
About Orthofix
Orthofix and SeaSpine merged in January 2023 to form a leading global spine and orthopedics company with a comprehensive portfolio of biologics, innovative spinal hardware, bone growth therapies, specialized orthopedic solutions and a leading surgical navigation system. Its products are distributed in approximately 68 countries worldwide.
The Company is headquartered in
Forward-Looking Statements
This news release may include forward-looking statements within the meaning of Section 21E of the Securities Exchange Act of 1934, as amended, and Section 27A of the Securities Act of 1933, as amended. In some cases, you can identify forward-looking statements by terminology such as “may,” “will,” “should,” “expects,” “plans,” “anticipates,” “believes,” “estimates,” “projects,” “intends,” “predicts,” “potential,” “continue” or other comparable terminology. Orthofix cautions you that statements included in this news release that are not a description of historical facts are forward-looking statements that are based on the Company’s current expectations and assumptions. Each forward-looking statement contained in this news release is subject to risks and uncertainties that could cause actual results to differ materially from those expressed or implied by such statement. Applicable risks and uncertainties include, among others: the ability of newly launched products to perform as designed and intended and to meet the needs of surgeons and patients, including as a result of the lack of robust clinical validation; and the risks identified under the heading “Risk Factors” in Orthofix Medical Inc.’s Annual Report on Form 10-K for the fiscal year ended December 31, 2022, which was filed with the Securities and Exchange Commission (SEC) on March 6, 2023. The Company’s public filings with the Securities and Exchange Commission are available at www.sec.gov. You are cautioned not to place undue reliance on forward-looking statements, which speak only as of the date when made. Orthofix does not intend to revise or update any forward-looking statement set forth in this news release to reflect events or circumstances arising after the date hereof, except as may be required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20231012672479/en/
Media Relations
Denise Landry
DeniseLandry@orthofix.com
214.937.2529
Investor Relations
Louisa Smith, Gilmartin Group
IR@orthofix.com
Source: Orthofix Medical Inc.
FAQ
What is the product being launched by Orthofix Medical Inc.?
What are the key features of OsteoCove?







