Additional Positive Data from Pivotal ASPEN Study of Brensocatib in Patients with Bronchiectasis to be Presented at the 7th World Bronchiectasis Conference
Insmed announced additional positive results from the ASPEN study on brensocatib at the 7th World Bronchiectasis Conference. The study assessed efficacy, safety, and tolerability of brensocatib in non-cystic fibrosis bronchiectasis. Key findings include a 21.1% risk reduction in pulmonary exacerbations for the 10 mg group and 19.4% for the 25 mg group, compared to placebo. The 25 mg dose also showed significantly less decline in lung function (FEV1) and forced vital capacity (FVC). Both doses improved Quality of Life scores, with nominally significant improvements in the 25 mg group. Brensocatib demonstrated a favorable safety profile, with common adverse events including COVID-19 and nasopharyngitis. Insmed plans to file a New Drug Application with the FDA in Q4 2024, with a potential U.S. launch in mid-2025, followed by Europe and Japan in 2026.
- 21.1% risk reduction in pulmonary exacerbations for brensocatib 10 mg (p=0.0019)
- 19.4% risk reduction in pulmonary exacerbations for brensocatib 25 mg (p=0.0046)
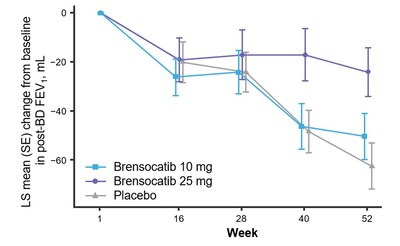
- Significantly less decline in FEV1 for brensocatib 25 mg (LS mean change of 38 mL, p=0.0054)
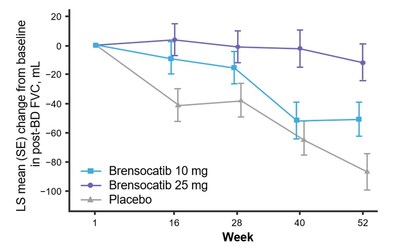
- Significantly less decline in FVC for brensocatib 25 mg (LS mean change of 75 mL, p<0.0001)
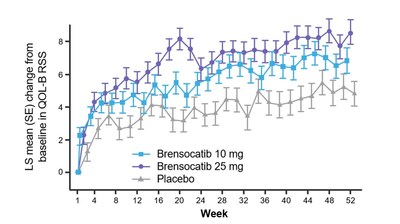
- Improvement in Quality of Life-Bronchiectasis Respiratory Symptom Domain Score for brensocatib 25 mg (3.8 points, p=0.0004)
- Brensocatib demonstrated a favorable safety profile
- New Drug Application to be filed with the FDA in Q4 2024
- Potential U.S. launch in mid-2025 and Europe/Japan in 2026
- None.
Insights
The recent data from the ASPEN study on brensocatib offers significant insights into its potential effectiveness and safety profile for treating non-cystic fibrosis bronchiectasis. Brensocatib's ability to reduce pulmonary exacerbations by 21.1% and 19.4% for the 10 mg and 25 mg doses respectively is noteworthy. These reductions are clinically meaningful considering that pulmonary exacerbations can severely impact the quality of life and health outcomes for patients.
Additionally, the less pronounced decline in lung function parameters such as FEV1 and FVC further supports the drug's potential benefits. A decline in FEV1 of 38 mL less than placebo is particularly promising because it indicates slower disease progression, which is important in chronic conditions like bronchiectasis.
The potential for brensocatib to be the first approved treatment for bronchiectasis also highlights the unmet need in this therapeutic area. This underscores the importance of advancing the drug through regulatory channels swiftly while ensuring robust safety and efficacy data.
Overall, the data is compelling for stakeholders, as it presents a balanced view of the benefits and manageable adverse events.
From a financial perspective, the positive outcomes of the ASPEN study are likely to have a significant impact on Insmed's market valuation. The anticipated filing with the FDA for a New Drug Application (NDA) by Q4 2024, followed by potential market approval and launches in key regions like the U.S., Europe and Japan, could drive substantial revenue growth for the company.
The fact that brensocatib could become the first approved treatment for bronchiectasis and the first dipeptidyl peptidase 1 (DPP1) inhibitor adds a strong competitive advantage. This could translate into a premium pricing strategy and significant market share, given the lack of existing treatments.
Investors should, however, consider the regulatory risks and the cost of commercialization. While the clinical data is promising, the path to regulatory approval can be complex and unpredictable. Additionally, the company's ability to effectively market and distribute the drug in various regions will be critical to its financial success.
In the short term, the positive clinical data can boost investor confidence and stock prices. In the long term, successful market penetration and financial returns will depend on regulatory outcomes and commercial execution.
The announcement of additional positive data from the ASPEN study is likely to enhance the competitive positioning of Insmed within the biopharmaceutical sector. The focus on a patient population with no currently approved treatments positions brensocatib to potentially fill a significant gap in the market.
The reported improvements in quality of life metrics such as the QOL-B Respiratory Symptom Domain Score and the BEST score are particularly noteworthy. These patient-reported outcomes are increasingly important in drug approval processes and can influence prescribing behaviors and patient adherence once the drug is on the market.
Moreover, the safety profile, comparable to placebo, suggests that brensocatib may be well-received by both physicians and patients, further driving its market potential.
Finally, the timelines for regulatory filings and potential market launches in the U.S., Europe and Japan are strategic, aiming to capitalize on market opportunities quickly. The staggered launches also allow for building on initial market experiences and tailoring strategies for subsequent regions.
This positions Insmed well for both short-term gains and long-term sustainability in the competitive landscape.
—New Graph Depicting 52-Week FEV1 Data Illustrate Significantly Less Decline in Lung Function for Brensocatib 25 mg Versus Placebo—
—Additional Exploratory Endpoints to be Presented, Including FVC and Patient-Reported BEST Score—
As previously announced, the
"The
The study assessed change in lung function, as measured by change from baseline in post-bronchodilator forced expiratory volume over one second (FEV1) at Week 52, a key secondary endpoint. Patients treated with brensocatib 25 mg demonstrated significantly less FEV1 decline, preserving more lung function as compared to placebo (LS mean change of 38 mL, p=0.0054). Patients in the placebo arm lost on average 62 mL of FEV1 in one year. In addition, new data will be presented at WBC measuring the change from baseline in post-bronchodilator forced vital capacity (FVC) at Week 52, another measure of lung function and an exploratory endpoint in the study. Patients treated with brensocatib 25 mg showed nominally significantly less decline in FVC compared to placebo (LS mean change of 75 mL, p<0.0001).
"We are incredibly excited to share additional results from the
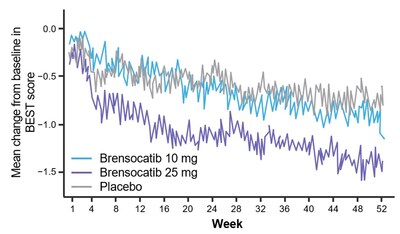
Patients in both dosage groups of brensocatib experienced numerical improvements in change from baseline in the Quality of Life-Bronchiectasis (QOL-B) Respiratory Symptom Domain Score, with the brensocatib 25 mg dose group demonstrating a nominally significant improvement of 3.8 points versus placebo (p=0.0004). Improvements in patient reported QOL-B Respiratory Symptom Domain Score were seen as early as 4 weeks in both brensocatib arms. New data will also be presented at WBC on the change in average daily bronchiectasis exacerbation and symptom tool (BEST) score, an exploratory endpoint, which is a novel symptom diary for bronchiectasis symptom burden and detection of exacerbations. Patients treated with brensocatib 25 mg showed a nominally significant 1-point decrease in BEST score compared to placebo.
Brensocatib was well-tolerated in the study and demonstrated a favorable safety profile. Treatment-emergent adverse events (TEAEs) occurring in at least
Insmed plans to file a New Drug Application with the
About
As part of the
About Bronchiectasis
Bronchiectasis is a serious, chronic lung disease in which the bronchi become permanently dilated due to a cycle of infection, inflammation, and lung tissue damage. The condition is marked by frequent pulmonary exacerbations requiring antibiotic therapy and/or hospitalizations. Symptoms include chronic cough, excessive sputum production, shortness of breath, and repeated respiratory infections, which can worsen the underlying condition. Bronchiectasis affects approximately 500,000 patients in the
About Brensocatib
Brensocatib is a small molecule, oral, reversible inhibitor of dipeptidyl peptidase 1 (DPP1) being developed by Insmed for the treatment of patients with bronchiectasis, CRSsNP, and other neutrophil-mediated diseases. DPP1 is an enzyme responsible for activating neutrophil serine proteases (NSPs), such as neutrophil elastase, in neutrophils when they are formed in the bone marrow. Neutrophils are the most common type of white blood cell and play an essential role in pathogen destruction and inflammatory mediation. In chronic inflammatory lung diseases, neutrophils accumulate in the airways and result in excessive active NSPs that cause lung destruction and inflammation. Brensocatib may decrease the damaging effects of inflammatory diseases such as bronchiectasis by inhibiting DPP1 and its activation of NSPs. Brensocatib is an investigational drug product that has not been approved for any indication in any jurisdiction.
About Insmed
Insmed Incorporated is a global biopharmaceutical company on a mission to transform the lives of patients with serious and rare diseases. Insmed's first commercial product is a first-in-disease therapy approved in the United States, Europe, and Japan to treat a chronic, debilitating lung disease. The Company is progressing a robust pipeline of investigational therapies targeting areas of serious unmet need, including neutrophil-mediated inflammatory diseases and rare pulmonary disorders. Insmed is also advancing an early-stage research engine encompassing a wide range of technologies and modalities, including artificial intelligence-driven protein engineering, gene therapy, and protein manufacturing. Insmed is headquartered in Bridgewater, New Jersey, with additional offices and research locations throughout the United States, Europe, and Japan. Visit www.insmed.com to learn more.
Forward-Looking Statements
This press release contains forward-looking statements that involve substantial risks and uncertainties. "Forward-looking statements," as that term is defined in the Private Securities Litigation Reform Act of 1995, are statements that are not historical facts and involve a number of risks and uncertainties. Words herein such as "may," "will," "should," "could," "would," "expects," "plans," "anticipates," "believes," "estimates," "projects," "predicts," "intends," "potential," "continues," and similar expressions (as well as other words or expressions referencing future events, conditions or circumstances) may identify forward-looking statements.
The forward-looking statements in this press release are based upon the Company's current expectations and beliefs, and involve known and unknown risks, uncertainties and other factors, which may cause the Company's actual results, performance and achievements and the timing of certain events to differ materially from the results, performance, achievements or timings discussed, projected, anticipated or indicated in any forward-looking statements. Such risks, uncertainties and other factors include, among others, the following: the risk that the full data set from the
The Company may not actually achieve the results, plans, intentions or expectations indicated by the Company's forward-looking statements because, by their nature, forward-looking statements involve risks and uncertainties because they relate to events and depend on circumstances that may or may not occur in the future. For additional information about the risks and uncertainties that may affect the Company's business, please see the factors discussed in Item 1A, "Risk Factors," in the Company's Annual Report on Form 10-K for the year ended December 31, 2023 and any subsequent Company filings with the Securities and Exchange Commission (SEC).
The Company cautions readers not to place undue reliance on any such forward-looking statements, which speak only as of the date of this press release. The Company disclaims any obligation, except as specifically required by law and the rules of the SEC, to publicly update or revise any such statements to reflect any change in expectations or in events, conditions or circumstances on which any such statements may be based, or that may affect the likelihood that actual results will differ from those set forth in the forward-looking statements.
Contact:
Investors:
Bryan Dunn
Executive Director, Investor Relations
Insmed
(646) 812-4030
bryan.dunn@insmed.com
Eleanor Barisser
Associate Director, Investor Relations
Insmed
(718) 594-5332
eleanor.barisser@insmed.com
Media:
Mandy Fahey
Executive Director, Corporate Communications
Insmed
(732) 718-3621
amanda.fahey@insmed.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/additional-positive-data-from-pivotal-aspen-study-of-brensocatib-in-patients-with-bronchiectasis-to-be-presented-at-the-7th-world-bronchiectasis-conference-302188927.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/additional-positive-data-from-pivotal-aspen-study-of-brensocatib-in-patients-with-bronchiectasis-to-be-presented-at-the-7th-world-bronchiectasis-conference-302188927.html
SOURCE Insmed Incorporated
FAQ
What are the results of the ASPEN study on brensocatib in bronchiectasis?
When will Insmed file for FDA approval of brensocatib?
What are the potential launch dates for brensocatib?
What are the safety findings of the ASPEN study for brensocatib?