Corvus Pharmaceuticals Announces Data from Cohort 2 of Placebo-Controlled Phase 1 Clinical Trial of Soquelitinib for Atopic Dermatitis
Corvus Pharmaceuticals (NASDAQ: CRVS) has released new interim data from its Phase 1 clinical trial of soquelitinib for atopic dermatitis. The trial, which includes data from 26 patients across two cohorts, showed promising results with 26% of treated patients achieving IGA 0 or 1 and 37% achieving EASI 75, while no placebo patients reached these endpoints.
The study evaluated two dosing regimens: Cohort 1 with 100 mg twice daily (12 patients) and Cohort 2 with 200 mg once daily (7 patients), against placebo groups. All Cohort 2 patients completed the 28-day treatment period with no significant safety issues or laboratory abnormalities. The trial is designed to enroll 64 patients total across four cohorts, with full results expected in Q2 2025.
Corvus Pharmaceuticals (NASDAQ: CRVS) ha rilasciato nuovi dati provvisori dal suo studio clinico di Fase 1 su soquelitinib per la dermatite atopica. Lo studio, che include dati provenienti da 26 pazienti suddivisi in due coorti, ha mostrato risultati promettenti con il 26% dei pazienti trattati che ha raggiunto IGA 0 o 1 e il 37% ha raggiunto EASI 75, mentre nessun paziente del gruppo placebo ha raggiunto questi obiettivi.
Lo studio ha valutato due regimi di dosaggio: Coorte 1 con 100 mg due volte al giorno (12 pazienti) e Coorte 2 con 200 mg una volta al giorno (7 pazienti), rispetto ai gruppi placebo. Tutti i pazienti della Coorte 2 hanno completato il periodo di trattamento di 28 giorni senza problemi di sicurezza significativi o anomalie di laboratorio. Lo studio è progettato per arruolare un totale di 64 pazienti in quattro coorti, con risultati completi previsti nel secondo trimestre del 2025.
Corvus Pharmaceuticals (NASDAQ: CRVS) ha publicado nuevos datos interinos de su ensayo clínico de Fase 1 sobre soquelitinib para la dermatitis atópica. El ensayo, que incluye datos de 26 pacientes en dos cohortes, mostró resultados prometedores con el 26% de los pacientes tratados alcanzando IGA 0 o 1 y el 37% alcanzando EASI 75, mientras que ningún paciente en el grupo placebo logró estos objetivos.
El estudio evaluó dos regímenes de dosis: Cohorte 1 con 100 mg dos veces al día (12 pacientes) y Cohorte 2 con 200 mg una vez al día (7 pacientes), frente a grupos placebo. Todos los pacientes de la Cohorte 2 completaron el período de tratamiento de 28 días sin problemas de seguridad significativos o anomalías de laboratorio. El ensayo está diseñado para inscribir un total de 64 pacientes en cuatro cohortes, con resultados completos esperados para el segundo trimestre de 2025.
코르부스 제약 (NASDAQ: CRVS)은 아토피 피부염 치료를 위한 soquelitinib의 1상 임상 시험에서 새로운 중간 데이터를 발표했습니다. 이 시험은 두 개의 코호트에서 26명의 환자의 데이터를 포함하고 있으며, 치료받은 환자의 26%가 IGA 0 또는 1에 도달하고 37%가 EASI 75를 달성하는 등 유망한 결과를 보였습니다. 반면, 플라세보 환자는 이러한 기준에 도달하지 못했습니다.
연구는 두 가지 용량 요법을 평가했습니다: 코호트 1은 하루 100mg를 두 번 (환자 12명), 코호트 2는 하루 200mg를 한 번 (환자 7명), 플라세보 그룹과 비교했습니다. 코호트 2의 모든 환자는 28일 치료 기간을 무사히 이수했으며, 안전성 문제나 실험실 이상 소견이 없었습니다. 이 시험은 총 64명의 환자를 네 개의 코호트에 등록할 계획이며, 전체 결과는 2025년 2분기에 예상됩니다.
Corvus Pharmaceuticals (NASDAQ: CRVS) a publié de nouvelles données intermédiaires de son essai clinique de phase 1 sur le soquelitinib pour la dermatite atopique. L'essai, qui comprend des données provenant de 26 patients répartis en deux cohortes, a montré des résultats prometteurs avec 26 % des patients traités atteignant IGA 0 ou 1 et 37 % atteignant EASI 75, alors qu'aucun patient du groupe placebo n'a atteint ces objectifs.
L'étude a évalué deux schémas posologiques : Cohorte 1 avec 100 mg deux fois par jour (12 patients) et Cohorte 2 avec 200 mg une fois par jour (7 patients), par rapport aux groupes placebo. Tous les patients de la Cohorte 2 ont complété la période de traitement de 28 jours sans problèmes de sécurité significatifs ni anomalies de laboratoire. L'essai est conçu pour enrôler un total de 64 patients répartis sur quatre cohortes, avec des résultats complets attendus au deuxième trimestre 2025.
Corvus Pharmaceuticals (NASDAQ: CRVS) hat neue Zwischenresultate aus seiner klinischen Phase-1-Studie zu Soquelitinib bei atopischer Dermatitis veröffentlicht. Die Studie, die Daten von 26 Patienten aus zwei Kohorten umfasst, zeigte vielversprechende Ergebnisse, wobei 26% der behandelten Patienten IGA 0 oder 1 und 37% EASI 75 erreichten, während keine Patienten in der Placebo-Gruppe diese Endpunkte erreichten.
Die Studie evaluierte zwei Dosierungsschemata: Kohorte 1 mit 100 mg zweimal täglich (12 Patienten) und Kohorte 2 mit 200 mg einmal täglich (7 Patienten) im Vergleich zu Placebo-Gruppen. Alle Patienten der Kohorte 2 schlossen die 28-tägige Behandlungsperiode ohne signifikante Sicherheitsprobleme oder Laboranomalien ab. Die Studie soll insgesamt 64 Patienten in vier Kohorten einbeziehen, wobei die vollständigen Ergebnisse für das zweite Quartal 2025 erwartet werden.
- 26% of treated patients achieved IGA 0 or 1 and 37% achieved EASI 75, while placebo group showed no improvements
- All Cohort 2 patients completed full 28-day treatment with no significant safety issues
- 200 mg once-daily dose (Cohort 2) showed better results than initial Cohort 1 results
- Trial is still in early Phase 1 stage, with only 26 out of planned 64 patients reported
- Full results not expected until Q2 2025
Insights
The interim Phase 1 data for soquelitinib shows compelling efficacy signals in atopic dermatitis treatment. The combined cohort analysis reveals that 26% of patients achieved IGA 0/1 and 37% achieved EASI 75, compared to zero response in the placebo group. These endpoints are particularly meaningful as they align with FDA's criteria for clinical significance and drug approval. The 200mg once-daily dosing in Cohort 2 appears to demonstrate superior outcomes compared to Cohort 1's twice-daily 100mg regimen, suggesting improved dosing optimization.
The safety profile remains clean with no significant adverse events or laboratory abnormalities, which is important for chronic conditions like atopic dermatitis where long-term treatment is often necessary. The oral administration route provides a significant competitive advantage over injectable alternatives in the market. The ongoing dose-escalation design with cohorts testing up to 400mg daily will help establish the optimal therapeutic window.
This data release strengthens Corvus's position in the $15 billion global atopic dermatitis market. The oral delivery format addresses a key market gap, as current leading treatments like Dupixent require injections. With positive efficacy signals and clean safety data, soquelitinib could capture significant market share, especially among patients seeking convenient oral alternatives.
The dual development strategy in both atopic dermatitis and T-cell lymphoma demonstrates smart pipeline diversification, potentially expanding the total addressable market. The upcoming J.P. Morgan Healthcare Conference presentation timing is strategic, potentially catalyzing investor interest. The planned Q2 2025 full data readout provides a near-term catalyst for stock movement.
Data continue to demonstrate a favorable safety and efficacy profile, including improvements in IGA 0 or 1 and EASI 75 scores compared to placebo
BURLINGAME, Calif., Jan. 13, 2025 (GLOBE NEWSWIRE) -- Corvus Pharmaceuticals, Inc. (NASDAQ: CRVS), a clinical-stage biopharmaceutical company, today announced new interim data from the randomized, double-blind, placebo-controlled Phase 1 clinical trial evaluating soquelitinib in patients with moderate to severe atopic dermatitis. The data, which include previously reported results from cohort 1 of the trial and new data covering 10 patients with 28-day follow-up from cohort 2 of the trial, demonstrated a favorable safety profile and efficacy profile. This includes significant responses in the soquelitinib treatment groups compared to placebo for clinically significant endpoints of IGA (Investigator Global Assessment) 0 or 1 and EASI (Eczema Area and Severity Index) 75.
“As we increase the number of patients, we remain excited by the outlook for our Phase 1 clinical trial of soquelitinib for the treatment of atopic dermatitis,” said Richard A. Miller, M.D., co-founder, president and chief executive officer of Corvus. “These additional data from cohort 2 evaluating a 200 mg once a day dose appear better than the initial results from cohort 1 reported in December. Using rigorous efficacy endpoints of IGA 0 or 1 and EASI 75, we see clinically meaningful activity of soquelitinib in both cohorts compared to the placebo group, which had no patients achieve these endpoints after four weeks of treatment. We focus on these endpoints because they have been accepted as measurements of clinical benefit and have been used as the basis for regulatory approval for several approved treatments for atopic dermatitis.”
Dr. Miller added, “Based on the results to-date, we believe soquelitinib will be attractive to physicians and patients due to its convenient oral administration, safety and novel mechanism of action. It also is well positioned as a potential new treatment option for a broad range of immune disease indications based on its ability to modulate and control parallel signaling pathways in the immune system. We look forward to completing enrollment in the atopic dermatitis trial and reporting full results in the second quarter 2025 while also continuing to advance our Phase 3 registration trial in relapsed peripheral T cell lymphoma and other clinical programs.”
Soquelitinib New Interim Data from the Atopic Dermatitis Phase 1 Clinical Trial
The Company is reporting results from 16 patients in Cohort 1 (12 patients in the soquelitinib group receiving 100 mg orally twice per day vs. four receiving placebo) and 10 patients in Cohort 2 (seven patients in the soquelitinib group receiving 200 mg orally once per day vs. three receiving placebo) for which 28 days of treatment have been completed. For those 19 patients in the soquelitinib group,
No significant safety issues were observed and no clinically significant laboratory abnormalities were seen. All 10 patients from Cohort 2 completed 28 days of dosing at the full dose of 200 mg orally once per day; the remaining patients from Cohort 2 are at various stages of treatment. Cohort 2 of the trial is fully enrolled (N=16) and the Company plans to report full results from all four study cohorts in the second quarter 2025.
Figure 1: Percent Patients Achieving Endpoints IGA 0 or 1, EASI 75 at Day 28 of Treatment
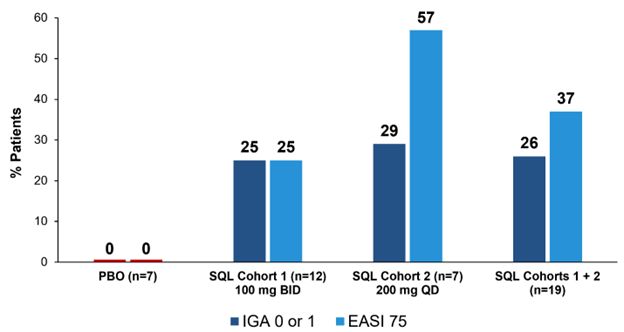
The placebo patients from Cohort 1 (n=4) and Cohort 2 (n=3) are combined.
Soquelitinib Atopic Dermatitis Phase 1 Clinical Trial Design
The randomized, double-blind, placebo-controlled Phase 1 clinical trial is planned to enroll 64 patients with moderate to severe atopic dermatitis that previously failed one prior topical or systemic therapy. Patients are enrolled into one of four dosing cohorts in a 3:1 ratio (12 active and four placebo) to receive either soquelitinib or placebo. The cohorts are sequentially enrolled and will examine 100 mg orally twice per day, 200 mg orally once per day, 200 mg orally twice per day and 400 mg orally once per day. Patients are treated for 28 days and are then followed for an additional 30 days with no therapy.
These doses were selected based on the Company’s prior experience evaluating soquelitinib in T cell lymphoma patients. The doses in the atopic dermatitis trial studied in Cohorts 1 and 2 are lower than the 200 mg orally twice a day dosing regimen, which is the level that has been shown to provide complete ITK occupancy and that is being evaluated in the Company’s ongoing registrational Phase 3 clinical trial of soquelitinib in peripheral T cell lymphoma.
The primary endpoints include safety and tolerability. Efficacy, measured by improvement in EASI score and IGA, are secondary endpoints. Reduction in itch and various cytokine biomarkers are exploratory endpoints. EASI scores are also evaluated by the percent of patients that achieve a specified percent reduction in EASI score – EASI 50 for patients that achieved a
Upcoming Presentation and Webcast
Dr. Miller will present a corporate overview including the new data from Cohort 2 of the soquelitinib Phase 1 clinical trial at the 43rd Annual J.P. Morgan Healthcare Conference on Wednesday, January 15 at 2:15 pm ET / 11:15 am PT. An audio webcast of the presentation will be available live and for 30 days following the event. The webcast may be accessed via the investor relations section of the Corvus website.
About Atopic Dermatitis
Atopic dermatitis, also called eczema, is a chronic disease that can cause inflammation, redness, scaly patches, blisters and irritation of the skin. It affects up to
About Soquelitinib
Soquelitinib (formerly CPI-818) is an investigational small molecule drug given orally designed to selectively inhibit ITK (interleukin-2-inducible T cell kinase), an enzyme that is expressed predominantly in T cells and plays a role in T cell and natural killer (NK) cell immune function. Soquelitinib has been shown to affect T cell differentiation and induce the generation of Th1 helper cells while blocking the development of both Th2 and Th17 cells and production of their secreted cytokines. Th1 T cells are required for immunity to tumors, viral infections and other infectious diseases. Th2 and Th17 helper T cells are involved in the pathogenesis of many autoimmune and allergic diseases. The Company believes the inhibition of specific molecular targets in T cells may be of therapeutic benefit for patients with cancers, including solid tumors, and in patients with autoimmune and allergic diseases. Recent studies have demonstrated that ITK controls a switch between the differentiation of Th17 proinflammatory cells and T regulatory suppressor cells. Inhibition of ITK leads to a shift toward T regulatory cell differentiation which has the potential to suppress autoimmune and inflammatory reactions. Based on interim results from a Phase 1/1b clinical trial in patients with refractory T cell lymphomas, which demonstrated tumor responses in very advanced, refractory, difficult to treat T cell malignancies, the Company has initiated a registrational Phase 3 clinical trial (NCT06561048) of soquelitinib in patients with relapsed PTCL. Soquelitinib is also now being investigated in a randomized placebo-controlled phase 1 clinical trial in patients with atopic dermatitis. A recent publication describing the chemistry, enzymology and biology of soquelitinib appeared in npj Drug Discovery in December 2024 and is available online at the Nature website and on the Publications and Presentations page of the Corvus website.
About Corvus Pharmaceuticals
Corvus Pharmaceuticals is a clinical-stage biopharmaceutical company pioneering the development of ITK inhibition as a new approach to immunotherapy for a broad range of cancer and immune diseases. The Company’s lead product candidate is soquelitinib, an investigational, oral, small molecule drug that selectively inhibits ITK. Its other clinical-stage candidates are being developed for a variety of cancer indications. For more information, visit www.corvuspharma.com.
Forward-Looking Statements
This press release contains forward-looking statements, including statements related to the potential safety and efficacy of the Company’s product candidates including soquelitinib; the outlook for the Phase 1 clinical trial of soquelitinib; physician and patient receptivity to soquelitinib; the potential use of soquelitinib to treat atopic dermatitis and other immune diseases; the Company’s conduct of, enrollment in and timing of clinical trials and results, including the Company’s Phase 3 clinical trial in PTCL and Phase 1 clinical trial in atopic dermatitis; and the potential of ITK inhibition as a new approach to immunotherapy. All statements other than statements of historical fact contained in this press release are forward-looking statements. These statements often include words such as “believe,” “expect,” “anticipate,” “intend,” “plan,” “estimate,” “seek,” “will,” “may” or similar expressions. Forward-looking statements are subject to a number of risks and uncertainties, many of which involve factors or circumstances that are beyond the Company’s control. The Company’s actual results could differ materially from those stated or implied in forward-looking statements due to a number of factors, including but not limited to, risks detailed in the Company’s Quarterly Report on Form 10-Q for the three months ended September 30, 2024, filed with the Securities and Exchange Commission on November 12, 2024, as well as other documents that may be filed by the Company from time to time with the Securities and Exchange Commission. In particular, the following factors, among others, could cause results to differ materially from those expressed or implied by such forward-looking statements: the Company’s ability to demonstrate sufficient evidence of efficacy and safety in its clinical trials of its product candidates; the accuracy of the Company’s estimates relating to its ability to initiate and/or complete preclinical studies and clinical trials and release data from such studies and clinical trials; the results of preclinical studies and interim data from clinical trials not being predictive of future results; the Company’s ability to enroll sufficient numbers of patients in its clinical trials; the unpredictability of the regulatory process; regulatory developments in the United States and other foreign countries; the costs of clinical trials may exceed expectations; and the Company’s ability to raise additional capital. Although the Company believes that the expectations reflected in the forward-looking statements are reasonable, it cannot guarantee that the events and circumstances reflected in the forward-looking statements will be achieved or occur, and the timing of events and circumstances and actual results could differ materially from those projected in the forward-looking statements. Accordingly, you should not place undue reliance on these forward-looking statements. All such statements speak only as of the date made, and the Company undertakes no obligation to update or revise publicly any forward-looking statements, whether as a result of new information, future events or otherwise.
INVESTOR CONTACT:
Leiv Lea
Chief Financial Officer
Corvus Pharmaceuticals, Inc.
+1-650-900-4522
llea@corvuspharma.com
MEDIA CONTACT:
Sheryl Seapy
Real Chemistry
+1-949-903-4750
sseapy@realchemistry.com
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/789edc2f-f3d8-4ffc-80c2-41765cceff02








