Cannabics Pharmaceuticals' Drug Candidate Exhibits 30% Tumor Volume Reduction in Mice
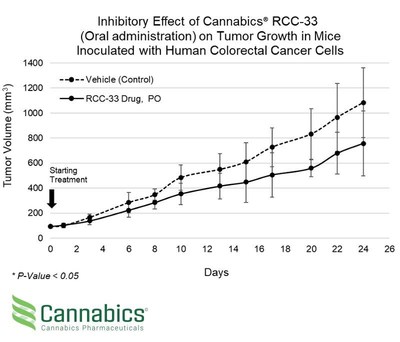
Cannabics Pharmaceuticals (OTCQB: CNBX) announced positive interim results from its second in-vivo POC study of drug candidate RCC-33, revealing a 30% reduction in tumor volume in colorectal cancer mice after 24 days of oral treatment. This follows prior results showing a 33% reduction via intraperitoneal injection. The study demonstrated statistical significance with a p-value under 0.05. Company executives emphasized progress towards human studies and enhancing drug bioavailability.
- 30% reduction in tumor volume in colorectal cancer mice after 24 days of oral RCC-33 treatment.
- Previous study showed a 33% reduction in tumor volume via intraperitoneal injection.
- Results indicate statistical significance with a p-value less than 0.05.
- Progress towards in-human studies and improving drug bioavailability.
- None.
Insights
Analyzing...
TEL AVIV, Israel and BETHESDA, Md., June 28, 2021 /PRNewswire/ -- Cannabics Pharmaceuticals Inc. (OTCQB: CNBX), a global leader in the development of cancer related cannabinoid-based medicine, released today the interim results of its second in-vivo POC study evaluating the efficacy of company's proprietary drug candidate RCC-33 for the treatment of colorectal cancer on mice. The study objective was to evaluate the potential efficacy of the RCC-33 drug candidate as a systemic treatment for colorectal cancer when administered orally. Interim study results confirmed the potential efficacy of RCC-33 as a systemic treatment for colorectal cancer when administered orally, showing a
The announcement comes following previously released in-vivo study results demonstrating a
Gabriel Yariv, Cannabics Pharmaceuticals President & COO said: "Our first POC study in mice demonstrated that RCC-33 has potential anti-tumor effect on colorectal cancer. During the first POC study we used intraperitoneal (IP) administration, via injection to the abdomen, and while we were very pleased to see a
Eyal Ballan Cannabics Pharmaceuticals CTO said "These positive results move us one step closer to in-human studies. After seeing the antitumor effect reconfirmed in this POC study, our next goal is to improve the bioavailability of the drug to be administered in our upcoming clinical studies".
About Cannabics Pharmaceuticals:
Cannabics Pharmaceuticals Inc. (OTCQB: CNBX) is a U.S. public company and a global leader in the development of cancer related cannabinoid-based medicine. The Company's R&D is based in Israel, where it is licensed by the Ministry of Health to conduct scientific and clinical research on cannabinoid formulations and cancer. For more information, please visit www.cannabics.com. For the latest updates on Cannabics Pharmaceuticals follow the Company on Twitter @Cannabics, Facebook @CannabicsPharmaceuticals, LinkedIn, and on Instagram @Cannabics_Pharmaceuticals.
Disclaimer:
Certain statements contained in this release may constitute forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995 and other U.S. Federal securities laws. Such statements include but are not limited to statements identified by words such as "believes," "expects," "anticipates," "estimates," "intends," "plans," "targets," "projects" and similar expressions. The statements in this release are based upon the current beliefs and expectations of our Company's management and are subject to significant risks and uncertainties. Actual results may differ from those outlined in the forward-looking statements. Numerous factors could cause or contribute to such differences, including, but not limited to, results of clinical trials and other studies, the challenges inherent in new product development initiatives, the effect of any competitive products, our ability to license and protect our intellectual property, our ability to raise additional capital in the future that is necessary to maintain our business, changes in government policy and regulation, potential litigation by or against us, any governmental review of our products or practices, as well as other risks discussed from time to time in our filings with the Securities and Exchange Commission including, without limitation, our latest 10-Q Report filed April 14th, 2021. We undertake no duty to update any forward-looking statement or any information contained in this press release or other public disclosures at any time. Finally, the investing public is reminded that the only announcements or information about Cannabics Pharmaceuticals Inc., which are condoned by the Company, must emanate from the Company itself and bear our name as its source.
For more information about Cannabics:
Cannabics Pharmaceuticals Inc.
Phone: +1-(877)-424-2429
info@Cannabics.com
http://www.Cannabics.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/cannabics-pharmaceuticals-drug-candidate-exhibits-30-tumor-volume-reduction-in-mice-301320931.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/cannabics-pharmaceuticals-drug-candidate-exhibits-30-tumor-volume-reduction-in-mice-301320931.html
SOURCE Cannabics Pharmaceuticals Inc.