PureTech Founded Entity Vedanta Biosciences Announces New Data from Phase 1 Study of VE202 for the Treatment of Inflammatory Bowel Disease
PureTech Health plc (Nasdaq: PRTC, LSE: PRTC) (“PureTech” or the “Company”), a clinical-stage biotherapeutics company dedicated to discovering, developing and commercializing highly differentiated medicines for devastating diseases, is pleased to note that its Founded Entity, Vedanta Biosciences, announced additional results from a Phase 1 study in healthy volunteers of VE202, Vedanta’s 16-strain live biotherapeutic product (LBP) candidate for inflammatory bowel disease (IBD). The new data were presented at the International Human Microbiome Consortium Congress 2021 (IHMC).
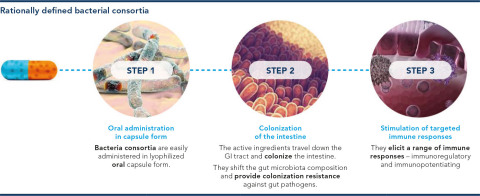
PureTech’s Founded Entity, Vedanta Biosciences, announced additional results from a Phase 1 study in healthy volunteers of VE202, Vedanta’s 16-strain live biotherapeutic product (LBP) candidate for inflammatory bowel disease (IBD). The new data were presented at the International Human Microbiome Consortium Congress 2021 (IHMC). (Photo: Business Wire)
Topline data from two Phase 1 studies exploring 11- and 16-strain VE202 consortia were announced in June 2020. The new data presented at IHMC summarized the long-term safety and colonization dynamics of the 16-strain version of VE202 in 31 healthy volunteers. Vedanta plans to move this consortium forward to a Phase 2 study in patients with ulcerative colitis. The study will be partially funded with proceeds from a
Key takeaways from the study include:
- All doses of the 16-strain consortium were generally safe and well-tolerated, with no reports of VE202-related serious adverse events.
- Multiple-day dosing proved superior to single-day dosing for inducing durable colonization; following 14 days of treatment, all or most of the LBP strains were detected in all VE202 recipients through the final study visit at Week 24.
- Colonization was abundant and durable; at the final study visit at Week 24, relative abundance of VE202 strains remained almost 100-fold higher than at baseline.
- Colonization was most effective when oral vancomycin pre-treatment preceded multiple doses of VE202.
- The results of this study corroborate the findings from other Phase 1 studies using Vedanta’s defined consortia and provide a framework to rationally select an optimal dose regimen for Phase 2 studies.
The full text of the announcement from Vedanta is as follows:
Vedanta Biosciences Presents New Data from Phase 1 Study of VE202, Its Rationally-Defined Consortium for the Treatment of Inflammatory Bowel Disease, at the International Human Microbiome Consortium Congress 2021 (IHMC)
Results further support the benign safety profile of VE202 and identify an optimal dosing regimen
Vedanta plans to initiate a Phase 2 clinical trial of VE202 in ulcerative colitis patients in the second half of 2021
CAMBRIDGE, MA, June 29, 2021 – Vedanta Biosciences, a leading clinical-stage microbiome company developing a new category of oral therapies using defined bacterial consortia manufactured from clonal cell banks, today announced additional results from a Phase 1 study in healthy volunteers of VE202, the Company’s 16-strain live biotherapeutic product (LBP) candidate for inflammatory bowel disease (IBD). The new data were presented at the International Human Microbiome Consortium Congress 2021 (IHMC), being held virtually from June 27-29th.
Topline data from two Phase 1 studies exploring 11- and 16-strain VE202 consortia were announced in June 2020. The new data presented at IHMC summarized the long-term safety and colonization dynamics of the 16-strain version of VE202 in 31 healthy volunteers. Vedanta plans to move this consortium forward to a Phase 2 study in patients with ulcerative colitis. The study will be partially funded with the proceeds from a
Key takeaways from the study include:
- All doses of the 16-strain consortium were generally safe and well-tolerated, with no reports of VE202-related serious adverse events.
- Multiple-day dosing proved superior to single-day dosing for inducing durable colonization; following 14 days of treatment, all or most of the LBP strains were detected in all VE202 recipients through the final study visit at Week 24.
- Colonization was abundant and durable; at the final study visit at Week 24, relative abundance of VE202 strains remained almost 100-fold higher than at baseline.
- Colonization was most effective when oral vancomycin pre-treatment preceded multiple doses of VE202.
- The results of this study corroborate the findings from other Phase 1 studies using Vedanta’s defined consortia and provide a framework to rationally select an optimal dose regimen for Phase 2 studies.
“Approximately 3 million people in the U.S. alone are affected by IBD, with another estimated 70,000 newly diagnosed cases each year. Although a number of treatments exist, they are limited and patients with IBD often struggle to find lasting relief with currently available medications. Our defined bacterial consortia are designed to reshape the ecosystem of the gut microbiome, an aspect of IBD that has not been addressed by biologics or other existing drug classes,” said Bernat Olle, Ph.D., co-founder and chief executive officer of Vedanta Biosciences. “We are encouraged by these results and look forward to advancing VE202 into Phase 2 clinical trials.”
Vedanta plans to initiate a Phase 2 clinical trial of VE202 in patients with mild to moderate ulcerative colitis in the second half of 2021.
About VE202
VE202 is a first-in-class, orally administered, investigational LBP consortium consisting of 16 bacterial strains of the Clostridia class, which were rationally selected to induce immune tolerance in the gut, reverse the gut microbiota abnormalities that are common in patients with IBD, and strengthen the epithelial barrier. Results describing the biology and candidate selection of VE202 were previously published in Science and Nature (multiple).
About Inflammatory Bowel Disease
Inflammatory bowel disease (IBD) is a debilitating, life-long condition that results from chronic inflammation of the intestinal tract. The most common forms of IBD are ulcerative colitis and Crohn's disease, which are both characterized by diarrhea, rectal bleeding, abdominal pain, fatigue and weight loss that can lead to life-threatening complications due to chronic inflammation of the digestive tract. Although the exact cause of IBD is not entirely understood, IBD is known to involve an interaction between genes, environmental factors, and dysregulation of the immune system, resulting in chronic inflammation. The CDC estimates that IBD affects an estimated
About Vedanta Biosciences
Vedanta Biosciences is leading the development of a potential new category of oral therapies for immune-mediated diseases using defined bacterial consortia manufactured from clonal cell banks. The Company’s approach bypasses the need to rely on direct sourcing of donor fecal material of inconsistent composition, thus overcoming challenges related to safety, quality, and scalability that limit donor-derived approaches. The clinical pipeline includes product candidates being evaluated for the treatment of C. difficile infection, inflammatory bowel diseases, advanced or metastatic cancers, and food allergy. These investigational therapies are grounded in pioneering research – published in leading journals including Science, Nature, and Cell – that identified bacteria that induce a range of beneficial immune responses. The Company’s platform includes what is believed to be the largest library of bacteria derived from the human microbiome, high-throughput methods for bacterial consortium design, vast datasets from human interventional studies, and state-of-the-art capabilities for cGMP-compliant manufacturing of defined bacterial consortia. Vedanta Biosciences controls a foundational intellectual property portfolio covering compositions of matter and methods of use for classes of bacteria that play key roles in human health. Vedanta Biosciences was founded by PureTech Health (Nasdaq: PRTC, LSE: PRTC) and a global team of scientific co-founders who pioneered the modern understanding of the interaction between the immune system and the microbiome.
About PureTech Health
PureTech is a clinical-stage biotherapeutics company dedicated to discovering, developing and commercializing highly differentiated medicines for devastating diseases, including inflammatory, fibrotic and immunological conditions, intractable cancers, lymphatic and gastrointestinal diseases and neurological and neuropsychological disorders, among others. The Company has created a broad and deep pipeline through the expertise of its experienced research and development team and its extensive network of scientists, clinicians and industry leaders. This pipeline, which is being advanced both internally and through PureTech's Founded Entities, is comprised of 26 therapeutics and therapeutic candidates, including two that have received FDA clearance and European marketing authorization, as of the date of PureTech’s most recently filed Annual Report on Form 20-F. All of the underlying programs and platforms that resulted in this pipeline of therapeutic candidates were initially identified or discovered and then advanced by the PureTech team through key validation points based on the Company's unique insights into the biology of the brain, immune and gut, or BIG, systems and the interface between those systems, referred to as the BIG Axis.
For more information, visit www.puretechhealth.com or connect with us on Twitter @puretechh.
Cautionary Note Regarding Forward-Looking Statements
This press release contains statements that are or may be forward-looking statements, including statements that relate to the company's future prospects, developments, and strategies. The forward-looking statements are based on current expectations and are subject to known and unknown risks and uncertainties that could cause actual results, performance and achievements to differ materially from current expectations, including, but not limited to, expectations regarding the potential of Vedanta’s VE202 for the treatment of IBD, expectations regarding the potential initiation and expected timing of a Phase 2 study of VE202 in ulcerative colitis, and those risks and uncertainties described in the risk factors included in the regulatory filings for PureTech Health plc. These forward-looking statements are based on assumptions regarding the present and future business strategies of the company and the environment in which it will operate in the future. Each forward-looking statement speaks only as at the date of this press release. Except as required by law and regulatory requirements, neither the company nor any other party intends to update or revise these forward-looking statements, whether as a result of new information, future events or otherwise.
View source version on businesswire.com: https://www.businesswire.com/news/home/20210630005198/en/







