Merck’s Clesrovimab (MK-1654), an Investigational Respiratory Syncytial Virus (RSV) Preventative Monoclonal Antibody, Significantly Reduced Incidence of RSV Disease and Hospitalization in Healthy Preterm and Full-term Infants
Merck (NYSE: MRK) announced positive results from the Phase 2b/3 clinical trial of clesrovimab, an investigational monoclonal antibody designed to protect infants from respiratory syncytial virus (RSV) disease. The trial met all prespecified endpoints, showing:
- 60.4% reduction in RSV-associated medically attended lower respiratory infections (MALRI)
- 84.2% reduction in RSV-associated hospitalizations
- 90.9% reduction in RSV-associated lower respiratory infection hospitalizations
- 91.7% reduction in severe MALRI
Clesrovimab demonstrated a comparable safety profile to palivizumab in a separate Phase 3 trial for at-risk infants. Merck aims to make clesrovimab available as early as the 2025-26 RSV season, potentially becoming the first immunization to protect both healthy and at-risk infants with a single dose regardless of weight.
Merck (NYSE: MRK) ha annunciato risultati positivi dallo studio clinico di fase 2b/3 di clesrovimab, un anticorpo monoclonale in fase di sperimentazione progettato per proteggere i neonati dalla malattia da virus sinciziale respiratorio (RSV). Lo studio ha raggiunto tutti gli obiettivi predefiniti, mostrando:
- Riduzione del 60,4% delle infezioni respiratorie inferiori mediche (MALRI) associate all'RSV
- Riduzione dell'84,2% dei ricoveri ospedalieri associati all'RSV
- Riduzione del 90,9% dei ricoveri ospedalieri per infezioni respiratorie inferiori associate all'RSV
- Riduzione del 91,7% delle MALRI gravi
Clesrovimab ha dimostrato un profilo di sicurezza comparabile a palivizumab in uno studio di fase 3 separato per neonati a rischio. Merck mira a rendere disponibile clesrovimab già dalla stagione RSV 2025-26, potenzialmente diventando la prima immunizzazione a proteggere sia i neonati sani che quelli a rischio con una sola dose indipendentemente dal peso.
Merck (NYSE: MRK) anunció resultados positivos del ensayo clínico de fase 2b/3 de clesrovimab, un anticuerpo monoclonal en investigación diseñado para proteger a los bebés de la enfermedad por virus sincitial respiratorio (RSV). El ensayo cumplió todos los objetivos predefinidos, mostrando:
- 60.4% de reducción en infecciones respiratorias inferiores atendidas médicamente asociadas al RSV (MALRI)
- 84.2% de reducción en hospitalizaciones asociadas al RSV
- 90.9% de reducción en hospitalizaciones por infecciones respiratorias inferiores asociadas al RSV
- 91.7% de reducción en MALRI severas
Clesrovimab demostró un perfil de seguridad comparable al palivizumab en un ensayo de fase 3 separado para bebés en riesgo. Merck planea hacer que clesrovimab esté disponible tan pronto como en la temporada de RSV 2025-26, convirtiéndose potencialmente en la primera inmunización para proteger tanto a los bebés sanos como a los de riesgo con una sola dosis, independientemente del peso.
머크 (NYSE: MRK)는 클레스로비맙의 2b/3상 임상 시험에서 긍정적인 결과를 발표했습니다. 이는 호흡기 세포 융합 바이러스 (RSV) 질환으로부터 유아를 보호하기 위해 설계된 연구용 단클론 항체입니다. 이 시험은 모든 사전 정의된 목표를 달성했으며, 다음과 같은 결과를 보였습니다:
- RSV 관련 의료적 치료를 받은 하부 호흡기 감염 (MALRI)에서 60.4% 감소
- RSV와 관련된 입원에서 84.2% 감소
- RSV와 관련된 하부 호흡기 감염으로 인한 입원에서 90.9% 감소
- 중증 MALRI에서 91.7% 감소
클레스로비맙은 위험이 있는 유아를 위한 별도의 3상 시험에서 팔리비주맙과 유사한 안전성 프로파일을 나타냈습니다. 머크는 클레스로비맙을 2025-26 RSV 시즌부터 제공할 계획이며, 이는 건강한 유아와 위험에 처한 유아 모두를 단일 용량으로 보호할 수 있는 첫 번째 예방접종이 될 가능성이 있습니다.
Merck (NYSE: MRK) a annoncé des résultats positifs de l'essai clinique de phase 2b/3 du clesrovimab, un anticorps monoclonal expérimental conçu pour protéger les nourrissons de la maladie par virus respiratoire syncytial (RSV). L'essai a atteint tous les objectifs prédéfinis, montrant :
- Réduction de 60,4 % des infections respiratoires inférieures médicalement assistées liées au RSV (MALRI)
- Réduction de 84,2 % des hospitalisations liées au RSV
- Réduction de 90,9 % des hospitalisations pour infections respiratoires inférieures liées au RSV
- Réduction de 91,7 % des MALRI sévères
Clesrovimab a démontré un profil de sécurité comparable à celui de palivizumab dans un essai de phase 3 séparé pour les nourrissons à risque. Merck vise à rendre le clesrovimab disponible dès la saison RSV 2025-26, devenant potentiellement la première immunisation à protéger à la fois les nourrissons en bonne santé et ceux à risque avec une seule dose, quel que soit leur poids.
Merck (NYSE: MRK) gab positive Ergebnisse der Phase 2b/3-Studie zu clesrovimab bekannt, einem experimentellen monoklonalen Antikörper, der entwickelt wurde, um Säuglinge vor der respiratorischen Synzytialvirus (RSV)-Erkrankung zu schützen. Die Studie erreichte alle vorab festgelegten Endpunkte und zeigte:
- 60,4% Reduktion der RSV-assoziierten medizinisch behandelten unteren Atemwegsinfektionen (MALRI)
- 84,2% Reduktion der RSV-assoziierten Krankenhausaufenthalte
- 90,9% Reduktion der RSV-assoziierten Krankenhausaufenthalte wegen niedriger Atemwegsinfektionen
- 91,7% Reduktion schwerer MALRI
Clesrovimab wies in einer separaten Phase-3-Studie für gefährdete Säuglinge ein vergleichbares Sicherheitsprofil zu Palivizumab auf. Merck beabsichtigt, Clesrovimab bereits in der RSV-Saison 2025-26 verfügbar zu machen, wodurch es möglicherweise die erste Impfung wird, die sowohl gesunde als auch gefährdete Säuglinge mit einer einzigen Dosis unabhängig vom Gewicht schützt.
- Clesrovimab reduced RSV-associated hospitalizations by 84.2%
- The drug reduced severe MALRI by 91.7%
- Clesrovimab showed a comparable safety profile to palivizumab in at-risk infants
- Potential to be the first immunization protecting both healthy and at-risk infants with a single dose
- None.
Insights
The Phase 2b/3 trial results for Merck's clesrovimab (MK-1654) are highly promising for RSV prevention in infants. Key findings include:
- 60.4% reduction in RSV-associated medically attended lower respiratory infections (primary endpoint)
- 84.2% reduction in RSV-associated hospitalizations
- 90.9% reduction in RSV-associated lower respiratory infection hospitalizations
- 91.7% reduction in severe MALRI
These results demonstrate significant clinical efficacy across various severity levels of RSV disease. The potential for a single dose regardless of infant weight could simplify administration compared to existing options. The comparable safety profile to palivizumab in high-risk infants further supports its potential. If approved, clesrovimab could become a game-changing preventive option for both healthy and at-risk infants, potentially reducing the substantial healthcare burden of RSV.
This news is highly positive for Merck (MRK) from a financial perspective. RSV is a significant market opportunity, with high infant hospitalization rates driving substantial healthcare costs. Key financial implications include:
- Potential first-mover advantage in the healthy infant RSV prevention market
- Simplified dosing could drive widespread adoption and market penetration
- Possible premium pricing given the strong efficacy data across disease severity levels
- Potential to capture market share from existing treatments like palivizumab in high-risk infants
With a target launch for the 2025-26 RSV season, Merck could see a significant revenue boost in the coming years. The addressable market is substantial, potentially encompassing millions of infants annually. This product could become a major growth driver for Merck's vaccine portfolio, diversifying revenue streams and strengthening the company's position in infectious disease prevention.
In the Phase 2b/3 trial, clesrovimab reduced RSV-associated hospitalizations (secondary endpoint) and RSV-associated lower respiratory infection hospitalizations (tertiary endpoint) by more than
Clesrovimab has the potential to become the first and only approved immunization designed to protect infants with the same single dose regardless of weight for the duration of their first RSV season
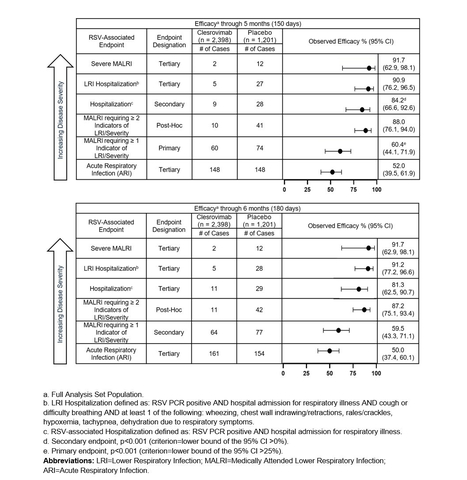
Figure 1 (Graphic: Business Wire)
Results from MK-1654-004, a placebo-controlled Phase 2b/3 pivotal trial evaluating a single dose of clesrovimab administered to healthy preterm and full-term infants (birth to 1 year of age) met all prespecified endpoints, with consistent results through both the 5-month and 6-month time points. The incidence of adverse events (AEs) and serious AEs were comparable between the clesrovimab and placebo groups, and there were no treatment or RSV-related deaths during the study.
“RSV continues to be a widespread seasonal infection that can affect both healthy and at-risk infants and is the leading cause of hospitalization for infants,” said Dr. Octavio Ramilo, chair of the Department of Infectious Diseases at St. Jude’s Children’s Research Hospital and investigator for the MK-1654-004 and MK-1654-007 trials. “The MK-1654-004 study evaluated a broad spectrum of RSV disease ranging from mild outpatient illness to severe disease requiring hospitalization. These promising results demonstrating decreased incidence of RSV disease, including hospitalizations, highlight the potential for clesrovimab to play an important role in helping to alleviate the continued burden of RSV on infants and their families.”
The primary efficacy endpoint of the trial, the reduction in incidence of RSV-associated medically attended lower respiratory infections (MALRI) requiring ≥ 1 indicator of lower respiratory infection (LRI) or severity compared to placebo through Day 150 (5 months) postdose, was
In addition, in a post hoc analysis, the reduction in incidence of MALRI requiring ≥ 2 indicators of LRI and severity (an endpoint of more severe MALRI than the primary MALRI endpoint), was
Additional details on the data from the MK-1654-004 trial across RSV disease burden are presented in order of decreasing disease severity endpoints in Figure 1 above.
Merck also announced data from a planned interim analysis of the MK-1654-007 trial, a Phase 3 trial evaluating the safety and efficacy of clesrovimab versus palivizumab in infants and children at increased risk for severe RSV disease. The primary endpoint of the study is the safety and tolerability of clesrovimab in infants entering their first RSV season. Interim results showed clesrovimab had a comparable safety profile to palivizumab, and no drug-related serious AEs were reported to date. Incidence rates of RSV-associated MALRI requiring ≥ 1 indicator of LRI or severity and RSV-associated hospitalizations (secondary endpoints) were also comparable between clesrovimab (
“The breadth of data presented at IDWeek highlight the potential for clesrovimab to help lessen the significant impact RSV can have on infants and their families, as well as the strain on healthcare systems due to high infection and hospitalization rates,” said Dr. Paula Annunziato, senior vice president, infectious diseases and vaccines, Global Clinical Development, Merck Research Laboratories. “These clinically meaningful findings also reinforce the potential for clesrovimab to be the first and only immunization designed to protect both healthy and at-risk infants using the same dose, regardless of weight. We look forward to continuing to discuss these data with health authorities around the world with the goal of making clesrovimab available for infants as early as the 2025-26 RSV season.”
About MK-1654-004
MK-1654-004 (NCT04767373) is a Phase 2b/3 double-blind, randomized, placebo-controlled clinical trial to evaluate the safety and efficacy of clesrovimab in healthy preterm and full-term infants from birth to 1 year of age entering their first RSV season. The study enrolled 3,632 participants who were randomized 2:1 to receive either a single fixed dose of clesrovimab (105 mg intramuscular injection (IM)) or placebo on Day 1. Primary endpoints included the incidence of participants with RSV-associated medically attended lower respiratory infection (MALRI) from Day 1 (postdose) to Day 150 as compared to placebo and safety. The MALRI definition required >1 indicator of LRI or severity. RSV-associated hospitalization through Day 150 and MALRI requiring >1 indicator of LRI or severity to Day 180, were prespecified secondary endpoints. Prespecified tertiary endpoints included acute respiratory infection, RSV-associated lower respiratory infection hospitalizations and incidence of severe MALRI through Day 150. In a post hoc analysis, more severe forms of RSV-associated MALRI (>2 indicators of LRI and severity) were assessed. Across endpoints, additional measures of efficacy were assessed through Day 180. Safety measures included the percentage of participants with solicited injection-related adverse events (AEs), AEs of special interest (AESIs) solicited systemic AEs or serious adverse events (SAEs).
About MK-1654-007
MK-1654-007 (NCT04938830) is a Phase 3, multicenter, randomized, partially blinded, controlled study to evaluate the safety, efficacy, and pharmacokinetics of clesrovimab in infants and children at increased risk for severe RSV disease compared to palivizumab. The study enrolled participants who were entering their first RSV season and recommended to receive palivizumab due to prematurity (≤35 weeks gestational age), chronic lung disease (CLD) of prematurity, or hemodynamically significant congenital heart disease (CHD). Participants were randomized 1:1 to receive clesrovimab (105 mg IM on Day 1, placebo on Day 28) or monthly palivizumab in their first season, and eligible participants received clesrovimab (210 mg IM) in the second RSV season. At this interim analysis, 901 participants were enrolled in the trial. The primary endpoint is safety and tolerability of clesrovimab versus palivizumab in the first season. Secondary endpoints include the incidence of RSV-associated medically attended lower respiratory infections (MALRI) requiring ≥1 indicator of LRI or severity and of RSV-associated hospitalization through Day 150.
About clesrovimab (MK-1654)
Clesrovimab (MK-1654) is an investigational, extended half-life monoclonal antibody (mAb) developed as a passive immunization for the prevention of RSV. Clesrovimab is designed to be administered as the same single dose, regardless of birth weight, and is being studied in healthy preterm, full-term and at-risk infants to provide direct, rapid, and durable protection through their first RSV season against mild, moderate and severe RSV.
About RSV
Respiratory syncytial virus (RSV) is a contagious virus that causes widespread seasonal infections like the flu, with a worldwide burden in infants and older adults. There is high unmet need for preventative options in both healthy and high-risk infants. Globally, RSV is the leading cause of hospitalization for healthy infants under a year old. RSV can lead to serious respiratory conditions like bronchiolitis and pneumonia, causing an estimated 101,000 deaths a year worldwide in children under five. According to the CDC, RSV season starts in the fall and peaks in the winter in most regions of
About Merck
At Merck, known as MSD outside of
Forward-Looking Statement of Merck & Co., Inc.,
This news release of Merck & Co., Inc.,
Risks and uncertainties include but are not limited to, general industry conditions and competition; general economic factors, including interest rate and currency exchange rate fluctuations; the impact of pharmaceutical industry regulation and health care legislation in
The company undertakes no obligation to publicly update any forward-looking statement, whether as a result of new information, future events or otherwise. Additional factors that could cause results to differ materially from those described in the forward-looking statements can be found in the company’s Annual Report on Form 10-K for the year ended December 31, 2023 and the company’s other filings with the Securities and Exchange Commission (SEC) available at the SEC’s Internet site (www.sec.gov).
View source version on businesswire.com: https://www.businesswire.com/news/home/20241017875997/en/
Media:
Julie Cunningham
(617) 519-6264
Kimberly Petrillo
(267) 742-2813
Investors:
Alexis Constantine
(732) 594-1578
Peter Dannenbaum
(732) 594-1579
Source: Merck & Co., Inc.







