INBRX-101 Shows Favorable Safety Profile in Patients with Alpha-1 Antitrypsin Deficiency and Demonstrates Potential to Achieve Normal Functional Alpha-1 Antitrypsin Levels with Monthly Dosing
Inhibrx Inc. announces favorable topline results from a Phase 1 clinical trial of INBRX-101, a recombinant human AAT-Fc fusion protein for treating alpha-1 antitrypsin deficiency (AATD). The study involved 31 patients and showed no severe drug-related adverse events, with mild and reversible issues reported. The drug achieved an average functional alpha-1 antitrypsin (AAT) level of 40.4 µM after the third dose, surpassing the current standard of care's 17.8 µM. Inhibrx believes INBRX-101 could reduce infusion frequency significantly, offering a more convenient treatment option.
- Topline results indicated a favorable safety and tolerability profile with no severe drug-related adverse events.
- INBRX-101 achieved a functional AAT level of 40.4 µM, higher than the standard of care level of 17.8 µM.
- Potential to reduce patient infusions from 52 to 12 times annually, improving patient convenience.
- None.
Insights
Analyzing...
- Topline results from the Phase 1 study showed a favorable safety and tolerability profile with no drug-related severe or serious adverse events.
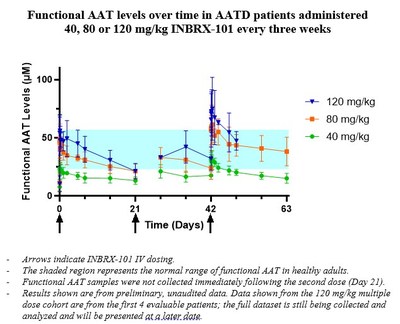
- Topline data from the multiple ascending dose cohorts of 40, 80 and 120 mg/kg demonstrated the average level ("Cavg") of functional alpha-1 antitrypsin ("AAT") achieved by INBRX-101 was 40.4 micromolar ("µM") over the 21-day dosing interval following the third 80 mg/kg dose.
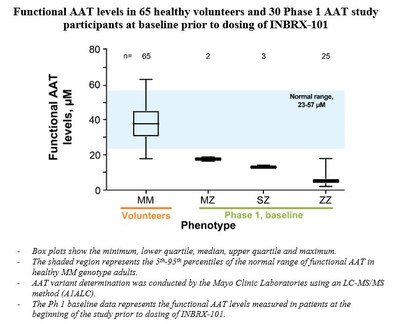
- Functional AAT levels collected from 65 healthy individuals with the MM genotype revealed a 5th/95th percentile range of 23 to 57 µM and a median of 38 µM.
SAN DIEGO, May 16, 2022 /PRNewswire/ -- Inhibrx Inc. (Nasdaq: INBX), a biotechnology company with four clinical programs in development and an emerging pre-clinical pipeline, today announced topline results from a Phase 1 clinical trial evaluating the safety, pharmacokinetics ("PK") and pharmacodynamics ("PD") of INBRX-101, an optimized recombinant human AAT-Fc fusion protein, in patients with alpha-1 antitrypsin deficiency ("AATD").
Data from this multi-country Phase 1 study are from 31 patients with AATD: 26 with the ZZ genotype, 3 with the SZ genotype and 2 with the MZ genotype of the SERPINA1 gene, the underlying cause of AATD. Treatment was well tolerated with no severe or serious adverse events related to the study drug. Drug-related adverse events were predominantly mild and those few that were moderate in severity were all transient and reversible, with minimal or no symptomatic care. No safety-related or PK/PD-related signs of neutralizing anti-drug antibodies were observed.
Dose-related increases in maximal and total INBRX-101 exposure occurred across the entirety of the single and multiple ascending dose ranges.
Data from the multiple ascending dose cohorts of INBRX-101 at 40, 80 and 120 mg/kg IV every three weeks showed the expected accumulation of functional AAT levels. Based on PK modeling, accumulation is expected to continue following subsequent doses and reach a steady-state after a total of approximately five to six consecutive doses, administered every three weeks.
The current standard of care, plasma-derived AAT, dosed once weekly at 60 mg/kg, achieves a Cavg of functional AAT of 17.8 µM over the weekly dosing interval as calculated from steady-state area under the curve ("AUC") values reported in Stocks et al. BMC Clinical Pharmacology 2010, 10:13. INBRX-101 achieved a mean Cavg of functional AAT of 40.4 µM over the 21-day dosing interval following the third 80 mg/kg dose.
To date, bronchoalveolar lavage fluid samples have been processed from two 80 mg/kg multiple ascending dose cohort individuals and confirm the presence of INBRX-101 in the lung fluid.
Additionally, functional AAT levels were measured in plasma samples from 65 normal MM genotype individuals. This analysis revealed the 5th and 95th percentiles of functional AAT levels in the normal MM genotype individuals were 23 and 57 µM, respectively, with a median of 38 µM.
"We believe this data demonstrates the potential of INBRX-101 to change the paradigm of treatment of AAT deficiency by maintaining patients in the normal range of functional AAT while reducing infusions from 52 annually to as few as 12 annually. We look forward to working with regulators, clinicians and patients to expedite this therapy to AAT deficient patients as rapidly as possible," said Mark Lappe, CEO of Inhibrx.
The Company will host a live webcast presentation today, May 16th, at 1:30 p.m. PT to further discuss the results.
Investors may join via the web: https://app.webinar.net/60dmpLaBwqx or may listen to the call by dialing (1-888-220-8451). Please refer to Inhibrx, Inc. or confirmation code 2516861 when calling in. Following the webcast, the presentation may be accessed through a link on the investors section of Inhibrx's website at https://inhibrx.investorroom.com/events-and-presentations. The webcast will be available for 60 days following the event. Following the presentation, Inhibrx will update its corporate presentation within the "Investors" section of its website at www.inhibrx.com.
INBRX-101 is a precisely engineered recombinant human AAT-Fc fusion protein designed to safely achieve and maintain levels of AAT found in healthy individuals with the potential for once-monthly dosing.
AATD is an inherited orphan disease affecting an estimated 100,000 patients in the United States. AATD is characterized by deficient levels of the AAT protein, which causes loss of lung tissue and function and decreased life expectancy. Plasma-derived AAT is the current standard of care and does not maintain patients in the normal AAT range, requires frequent and inconvenient once-weekly IV dosing, and relies on plasma collection practices that might not be sustainable.
Inhibrx is a clinical-stage biotechnology company focused on developing a broad pipeline of novel biologic therapeutic candidates in oncology and orphan diseases. Inhibrx utilizes diverse methods of protein engineering to address the specific requirements of complex target and disease biology, including its proprietary sdAb platform. Inhibrx has collaborations with 2seventy bio (formerly bluebird bio), Bristol-Myers Squibb and Chiesi. For more information, please visit www.inhibrx.com.
Inhibrx cautions you that statements contained in this press release regarding matters that are not historical facts are forward-looking statements. These statements are based on Inhibrx's current beliefs and expectations. These forward-looking statements include, but are not limited to, statements regarding: Inhibrx's and its investigators' judgments and beliefs regarding the observed safety and efficacy to date of its therapeutic candidate, INBRX-101, discussions with and beliefs regarding future action by the U.S. Food and Drug Administration, statements and beliefs regarding the current standard of care for AAT and the sustainability of current plasma collection practices and the potential for INBRX-101 to change the standard of care, future clinical development, application and dosage of INBRX-101 and the presumption that topline data will be representative of final data and that PK modeling is an accurate predictor of PK levels on a broader basis. Actual results may differ from those set forth in this press release due to the risks and uncertainties inherent in Inhibrx's business, including, without limitation, risks and uncertainties regarding: the initiation, timing, progress and results of its preclinical studies and clinical trials, and its research and development programs; its ability to advance therapeutic candidates into, and successfully complete, clinical trials; its interpretation of initial, interim or preliminary data from its clinical trials, including interpretations regarding disease control and disease response; the timing or likelihood of regulatory filings and approvals; the successful commercialization of its therapeutic candidates, if approved; the pricing, coverage and reimbursement of its therapeutic candidates, if approved; its ability to utilize its technology platform to generate and advance additional therapeutic candidates; the implementation of its business model and strategic plans for its business and therapeutic candidates; its ability to successfully manufacture therapeutic candidates for clinical trials and commercial use, if approved; its ability to contract with third-party suppliers and manufacturers and their ability to perform adequately; the scope of protection it is able to establish and maintain for intellectual property rights covering its therapeutic candidates; its ability to enter into strategic partnerships and the potential benefits of these partnerships; its estimates regarding expenses, capital requirements and needs for additional financing and financial performance; its expectations regarding the impact of the COVID-19 pandemic on its business; and other risks described in Inhibrx's filings with the U.S. Securities and Exchange Commission (the "SEC"), including under the heading "Risk Factors" in Inhibrx's Annual Report on Form 10-K for the year ended December 31, 2021, as filed with the SEC on February 28, 2022, as well as its Quarterly Reports on Form 10-Q, and supplemented from time to time by its Current Reports on Form 8-K. You are cautioned not to place undue reliance on these forward-looking statements, which speak only as of the date hereof, and Inhibrx undertakes no obligation to update these statements to reflect events that occur or circumstances that exist after the date hereof. All forward-looking statements are qualified in their entirety by this cautionary statement, which is made under the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. This press release contains estimates and other statistical data made by independent parties and by Inhibrx. This data involves a number of assumptions and limitations, and you are cautioned not to give undue weight to such estimates.
Investor and Media Contact:
Kelly Deck, CFO
kelly@inhibrx.com
858-795-4260
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/inbrx-101-shows-favorable-safety-profile-in-patients-with-alpha-1-antitrypsin-deficiency-and-demonstrates-potential-to-achieve-normal-functional-alpha-1-antitrypsin-levels-with-monthly-dosing-301547416.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/inbrx-101-shows-favorable-safety-profile-in-patients-with-alpha-1-antitrypsin-deficiency-and-demonstrates-potential-to-achieve-normal-functional-alpha-1-antitrypsin-levels-with-monthly-dosing-301547416.html
SOURCE Inhibrx Inc.