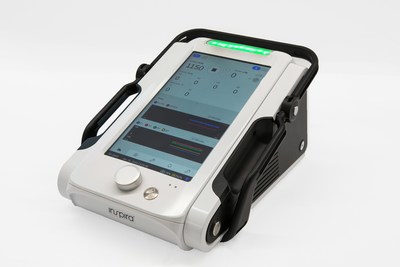
Inspira™ Technologies Begins Manufacturing of the ALICE™ CPB Device, ahead of planned 2023 FDA submission
Inspira Technologies (NASDAQ: IINN) has initiated manufacturing for the ALICE CPB device, crucial for its market strategy. This device is set for FDA 510(k) submission in 2023. The ALICE device aims to enhance CPB support in healthcare settings, equipped with a large touchscreen and real-time blood monitoring capabilities via the HYLA™ Blood Sensor. Contract manufacturing is conducted by a qualified provider meeting GMP standards. The company aims to improve patient care accessibility globally and is establishing partnerships for clinical adoption.
- Initiation of manufacturing for the ALICE device, key for market penetration.
- Planned submission for FDA 510(k) clearance in 2023.
- Potential advantages of the ALICE device in medical settings, including real-time blood monitoring.
- None.
Insights
Analyzing...
The ALICE device is an important part of Inspira's market penetration strategy.
RA'ANANA, Israel, Dec. 20, 2022 /PRNewswire/ -- Inspira Technologies OXY B.H.N. Ltd. (NASDAQ: IINN) (NASDAQ: IINNW) (the "Company" or "Inspira Technologies"), a groundbreaking respiratory support technology company, announced today that it has begun the manufacturing process for the ALICE CPB (Cardiopulmonary Bypass) device (the "ALICE device") to undergo the Verification and Validation phase prior to its planned 2023 submission to the U.S. Food and Drug Administration (FDA) for 510(k) clearance. If FDA clearance is obtained, the ALICE device production line will be extended for Low-Rate Initial Production (LWRIP), which is an important operational stage in developing infrastructure to support serial manufacturing, quality control and shipping. Additional units are expected to be assembled for the targeted future first deployments of the ALICE devices in the U.S. and Israel.
The ALICE device is being contract manufactured by an end-to-end solution provider that offers New Product Introduction (NPI) services to mass production capabilities for leading medical electronic device companies. This includes full turn-key manufacturing, full system integration, including PCB (Printed Circuit Board) manufacturing, assembly services, testing and packaging in facilities that meet Good Manufacturing Practices (GMP) compliance.
The ALICE device is intended to provide potential advantages in medical device design, including a large touchscreen and novel colorful graphical presentation, that increases the visibility and functionality of data displayed to the medical staff. The ALICE device is being designed to be both lightweight and highly durable and is designed to be equipped with long battery life. The ALICE device is intended to be indicated for use in clinical settings requiring CPB support. Inspira plans for the ALICE to be the first device to Integrate the HYLA™ Blood Sensor designed to be non-invasive, perform continuous measurements and alert physicians at real time of sudden changes in a patient's specific key blood indicators.
Inspira Technologies' goal is to set a new standard in various aspects of patient care, targeting intensive care units (ICUs), general medical units, operating theaters, emergency medical services and small urban and rural hospitals, with the focus of increasing accessibility to medical care for millions of patients worldwide. As part of our strategy to reach these goals, and in parallel to pursuing regulatory approvals, we are actively working to establish collaborations with strategic partners and globally ranked health centers to provide endorsement and clinical adoption for regional deployments of our respiratory support, CPB, and blood measuring products and technologies.
"Today, we believe that we have achieved a very important milestone in line with our strategy towards the production and delivery of Inspira Technologies' products to the market," stated Avi Shabtai, Inspira Technologies' Chief Operating Officer.
Inspira Technologies OXY B.H.N. Ltd.
Inspira Technologies is an innovative medical technology company in the respiratory treatment arena. The Company has developed a breakthrough Augmented Respiration Technology (INSPIRA ART), designed to rebalance patient oxygen saturation levels. This technology potentially allows patients to remain awake during treatment while minimizing the need for highly invasive, risky, and costly mechanical ventilation systems that require intubation and medically induced coma. The Company's products have not yet been tested or used in humans and has not been approved by any regulatory entity.
For more information, please visit our corporate website: https://Inspira-Technologies.com
Forward-Looking Statement Disclaimer
This press release contains express or implied forward-looking statements pursuant to U.S. Federal securities laws. These forward-looking statements and their implications are based on the current expectations of the management of the Company only and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. For example, the Company is using forward-looking statements when it discusses its regulatory strategy, the timing of the expected submission of the ALICE device for 510k clearance, the extension of indications and uses for its ALICE product following FDA clearance, its intended manufacturing plans and intended first targeted markets for the ALICE device, the potential advantages, and benefits to be derived by its products, its goal is to set a new standard in various aspects of patient care and increase accessibility to medical care for millions of patients worldwide, its pursuit of collaborations with strategic partners and globally ranked health centers and its belief that it has achieved a very important milestone in line with its strategy towards the production and delivery of its products to the market. These forward-looking statements and their implications are based solely on the current expectations of the Company's management and are subject to a number of factors and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as otherwise required by law, the Company undertakes no obligation to publicly release any revisions to these forward-looking statements to reflect events or circumstances after the date hereof or to reflect the occurrence of unanticipated events. More detailed information about the risks and uncertainties affecting the Company is contained under the heading "Risk Factors" in the Company's annual report on Form 20-F for the fiscal year ended December 31, 2021 filed with the U.S. Securities and Exchange Commission (the "SEC"), which is available on the SEC's website, www.sec.gov
For more details:
US Investor & Public Relations
Dave Gentry
RedChip Companies Inc.
1-800-RED-CHIP (733-2447)
Or 407-491-4498
IINN@redchip.com
Copyright © 2018-2022 Inspira Technologies OXY B.H.N. LTD., All rights reserved.
Photo: https://mma.prnewswire.com/media/1972158/ALICE_CPB_device.jpg
Logo: https://mma.prnewswire.com/media/1668495/Inspira_Technologies_Logo.jpg
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/inspira-technologies-begins-manufacturing-of-the-alice-cpb-device-ahead-of-planned-2023-fda-submission-301707255.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/inspira-technologies-begins-manufacturing-of-the-alice-cpb-device-ahead-of-planned-2023-fda-submission-301707255.html
SOURCE Inspira Technologies