Chemomab Reports Positive Results in Nebokitug Phase 2 PSC Open Label Extension Trial Showing Continued Broad and Substantial Improvements in Key Liver Biomarkers
Chemomab Therapeutics (Nasdaq: CMMB) has reported positive results from the Open Label Extension (OLE) portion of its Phase 2 SPRING trial for nebokitug in primary sclerosing cholangitis (PSC) patients. The trial showed continued improvements across key liver biomarkers after 48 weeks of treatment.
Key findings include:
- Over 90% of eligible SPRING trial patients participated in the OLE
- Patients with moderate/advanced disease showed significantly lower clinical events (4.8%) compared to historical controls (25.8%)
- Treatment demonstrated improvements in liver injury, inflammation, and fibrosis markers
- The drug was well-tolerated at both 10 mg/kg and 20 mg/kg doses administered every three weeks
The results support nebokitug's potential to become the first approved drug with disease-modifying activity for PSC, a deadly disorder currently lacking effective treatments. The 20 mg/kg dose showed particularly strong results and will be used in the upcoming Phase 3 trial.
Chemomab Therapeutics (Nasdaq: CMMB) ha riportato risultati positivi dalla parte di estensione in aperto (OLE) del suo studio di Fase 2 SPRING per nebokitug nei pazienti con colangite sclerosante primaria (PSC). Lo studio ha mostrato miglioramenti continui in vari biomarcatori epatici chiave dopo 48 settimane di trattamento.
I risultati principali includono:
- Oltre il 90% dei pazienti idonei allo studio SPRING ha partecipato all'OLE
- I pazienti con malattia moderata/avanzata hanno mostrato eventi clinici significativamente inferiori (4,8%) rispetto ai controlli storici (25,8%)
- Il trattamento ha dimostrato miglioramenti nei marcatori di danno epatico, infiammazione e fibrosi
- Il farmaco è stato ben tollerato sia alla dose di 10 mg/kg che a quella di 20 mg/kg somministrate ogni tre settimane
I risultati supportano il potenziale di nebokitug di diventare il primo farmaco approvato con attività modificante la malattia per la PSC, una malattia mortale attualmente priva di trattamenti efficaci. La dose di 20 mg/kg ha mostrato risultati particolarmente forti e sarà utilizzata nel prossimo studio di Fase 3.
Chemomab Therapeutics (Nasdaq: CMMB) ha reportado resultados positivos de la parte de Extensión Abierta (OLE) de su ensayo de Fase 2 SPRING para nebokitug en pacientes con colangitis esclerosante primaria (PSC). El ensayo mostró mejoras continuas en varios biomarcadores hepáticos clave después de 48 semanas de tratamiento.
Los hallazgos clave incluyen:
- Más del 90% de los pacientes elegibles del ensayo SPRING participaron en el OLE
- Los pacientes con enfermedad moderada/avanzada mostraron eventos clínicos significativamente más bajos (4.8%) en comparación con controles históricos (25.8%)
- El tratamiento demostró mejoras en los marcadores de daño hepático, inflamación y fibrosis
- El fármaco fue bien tolerado tanto a dosis de 10 mg/kg como de 20 mg/kg administradas cada tres semanas
Los resultados respaldan el potencial de nebokitug para convertirse en el primer fármaco aprobado con actividad modificadora de la enfermedad para PSC, un trastorno mortal que actualmente carece de tratamientos efectivos. La dosis de 20 mg/kg mostró resultados particularmente sólidos y se utilizará en el próximo ensayo de Fase 3.
Chemomab Therapeutics (Nasdaq: CMMB)는 주요 경화성 담관염(PSC) 환자를 위한 nebokitug의 2상 SPRING 시험의 공개 라벨 확장(OLE) 부분에서 긍정적인 결과를 보고했습니다. 이 시험은 48주 치료 후 주요 간 바이오마커에서 지속적인 개선을 보여주었습니다.
주요 발견 사항은 다음과 같습니다:
- SPRING 시험의 적격 환자 중 90% 이상이 OLE에 참여했습니다.
- 중등도/진행성 질환 환자는 역사적 대조군(25.8%)에 비해 임상적 사건이 유의미하게 낮았습니다(4.8%).
- 치료는 간 손상, 염증 및 섬유증 마커에서 개선을 보여주었습니다.
- 약물은 10 mg/kg 및 20 mg/kg 용량 모두에서 3주마다 투여 시 잘 견뎌졌습니다.
이 결과는 nebokitug가 PSC에 대한 질병 수정 활동을 가진 최초의 승인된 약물이 될 잠재력을 지지합니다. 20 mg/kg 용량은 특히 강력한 결과를 보여주었으며, 다음 3상 시험에서 사용될 예정입니다.
Chemomab Therapeutics (Nasdaq: CMMB) a rapporté des résultats positifs de la partie d'Extension en Ouvert (OLE) de son essai de Phase 2 SPRING pour le nebokitug chez les patients atteints de cholangite sclérosante primitive (PSC). L'essai a montré des améliorations continues dans plusieurs biomarqueurs hépatiques clés après 48 semaines de traitement.
Les principales conclusions incluent:
- Plus de 90 % des patients éligibles de l'essai SPRING ont participé à l'OLE
- Les patients atteints de maladie modérée/avancée ont montré des événements cliniques significativement plus bas (4,8 %) par rapport aux contrôles historiques (25,8 %)
- Le traitement a montré des améliorations dans les marqueurs de lésions hépatiques, d'inflammation et de fibrose
- Le médicament a été bien toléré à la fois à des doses de 10 mg/kg et de 20 mg/kg administrées toutes les trois semaines
Les résultats soutiennent le potentiel du nebokitug à devenir le premier médicament approuvé avec une activité modifiant la maladie pour la PSC, un trouble mortel actuellement dépourvu de traitements efficaces. La dose de 20 mg/kg a montré des résultats particulièrement solides et sera utilisée dans l'essai de Phase 3 à venir.
Chemomab Therapeutics (Nasdaq: CMMB) hat positive Ergebnisse aus dem Open Label Extension (OLE)-Teil seiner Phase-2-SPRING-Studie für Nebokitug bei Patienten mit primärer sklerosierender Cholangitis (PSC) berichtet. Die Studie zeigte nach 48 Wochen Behandlung fortlaufende Verbesserungen in wichtigen Leber-Biomarkern.
Wichtige Ergebnisse umfassen:
- Über 90 % der geeigneten Patienten der SPRING-Studie nahmen am OLE teil
- Patienten mit moderater/fortgeschrittener Erkrankung wiesen signifikant niedrigere klinische Ereignisse (4,8 %) im Vergleich zu historischen Kontrollen (25,8 %) auf
- Die Behandlung zeigte Verbesserungen bei Leberverletzungen, Entzündungen und Fibrose-Markern
- Das Medikament wurde sowohl in Dosen von 10 mg/kg als auch 20 mg/kg, die alle drei Wochen verabreicht wurden, gut vertragen
Die Ergebnisse unterstützen das Potenzial von Nebokitug, das erste zugelassene Medikament mit krankheitsmodifizierender Wirkung für PSC zu werden, eine tödliche Erkrankung, für die derzeit effektive Behandlungen fehlen. Die Dosis von 20 mg/kg zeigte besonders starke Ergebnisse und wird in der kommenden Phase-3-Studie verwendet.
- Significant reduction in clinical events: 4.8% in treated patients vs 25.8% in historical controls
- Continued improvements in key liver biomarkers over 48 weeks
- High patient retention with 90%+ eligible patients continuing treatment
- Strong safety profile maintained over extended 48-week period
- Positive results particularly strong in planned Phase 3 dose (20 mg/kg)
- Drug still requires Phase 3 trial completion before potential approval
- Results based on comparison to historical controls rather than concurrent placebo group
Insights
Chemomab's latest results from the 48-week Open Label Extension (OLE) of their Phase 2 SPRING trial for nebokitug significantly strengthen the drug's clinical profile in Primary Sclerosing Cholangitis (PSC), a deadly liver condition with no FDA-approved treatments.
The data shows continued improvements across key liver biomarkers of injury, inflammation, and fibrosis - particularly in ELF score components and PRO-C3 levels. Most notably, patients receiving nebokitug for 48 weeks experienced significantly fewer clinical events (
These findings are especially important because they directly derisk the planned Phase 3 trial design, where clinical events will serve as the primary endpoint. The strong results in the 20 mg/kg dose group (selected for Phase 3) and in patients with moderate/advanced disease (the Phase 3 target population) further strengthen the clinical development pathway.
The high participation rate (over
These Phase 2 OLE results represent a substantial positive development for Chemomab's lead asset nebokitug. The data meaningfully derisks the clinical program by demonstrating extended durability of treatment effect over 48 weeks, a critical factor for a chronic progressive disease like PSC.
The significant reduction in clinical events (
Nebokitug's competitive positioning looks increasingly strong. With no approved therapies for PSC, the first effective treatment could capture substantial market share. The comprehensive biomarker improvements across multiple aspects of the disease (liver injury, inflammation, fibrosis) suggest a potential mechanism that addresses the underlying pathology rather than merely treating symptoms.
For a company with a market cap of just
Reinforces potential of nebokitug to become the first approved drug with disease-modifying activity for PSC--a deadly disorder that currently has no effective treatments
Treatment in PSC patients with moderate/advanced disease for 48 weeks resulted in continued improvements across key biomarkers of liver injury, inflammation and fibrosis, including ELF score, fibrosis-related ELF score components and PRO-C3
Patients treated with nebokitug for 48 weeks showed a significantly lower number of clinical events compared to matching historical controls and significantly reduced progression of liver stiffness vs historical controls, further derisking the planned PSC Phase 3 clinical trial design
Treatment for up to 48 weeks was well-tolerated; Reinforces and expands positive results from 15-week double-blind Phase 2 SPRING trial
TEL AVIV, Israel, March 27, 2025 (GLOBE NEWSWIRE) -- Chemomab Therapeutics, Ltd., (Nasdaq: CMMB), a clinical stage biotechnology company developing innovative therapeutics for fibro-inflammatory diseases with high unmet need, today announced positive results from the Open Label Extension (OLE) portion of the Phase 2 SPRING trial assessing nebokitug as a treatment for patients with primary sclerosing cholangitis (PSC). More than
“We believe these positive results represent an important milestone for nebokitug, providing further clinical proof-of-concept in PSC and derisking our Phase 3 program. They are great news for Chemomab and for patients with PSC, a debilitating disorder that lacks any FDA-approved treatments and too often results in liver transplantation or death,” said Adi Mor, PhD, co-founder and Chief Executive Officer of Chemomab. “Since PSC is a chronic, slowly progressive disease, it was important to demonstrate that the positive results reported from the 15-week double-blind Phase 2 study were durable over a longer period of treatment. We are delighted that nebokitug continued to be safe and well-tolerated over 12 months of treatment and showed broad and substantial improvements in all the key biomarkers associated with PSC. The results were especially strong in the 20 mg/kg dose that we expect to use in the Phase 3 trial, as well as in patients with moderate/advanced disease, a group that is most at risk for disease progression and that will be the focus in Phase 3.”
The OLE study confirmed that the drug was safe and well-tolerated in PSC patients receiving 10 mg/kg or 20 mg/kg of nebokitug administered once every three weeks for up to 48 weeks, and resulted in positive effects, including continued improvements in key liver biomarkers such as the ELF score, the fibrosis-related components of ELF and the fibrosis biomarker PRO-C3. Liver stiffness scores (transient elastography), as measured by FibroScan® were substantially lower in the nebokitug-treated patients with moderate/advanced disease compared to matching historical controls. Cholestasis-related markers stabilized over 48 weeks of treatment and total serum bile acids were reduced. OLE patients with moderate/advanced disease treated with nebokitug for 48 weeks showed a significantly lower number of clinical events (
Nebokitug Treatment for 48 Weeks Stabilizes Risk of Disease Progression as Determined by ELF Score
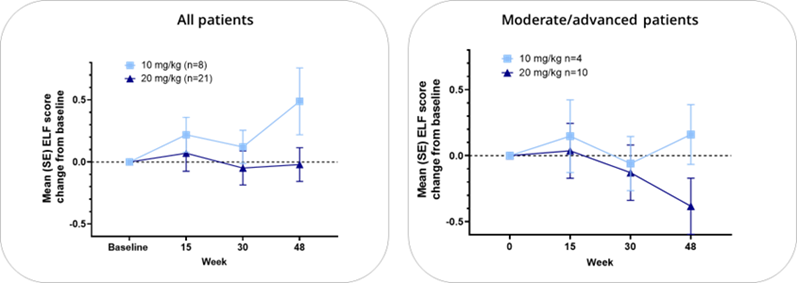
All Patients-All OLE completers; Moderate/advanced by ELF (ELF>9.8 at baseline)
Nebokitug Treatment for 48 Weeks Slowed Progression of Liver Stiffness Compared to Historical Controls
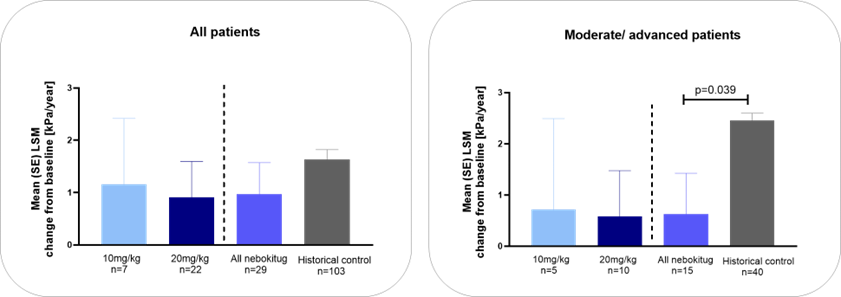
Moderate/Advanced by LSM (F2-F3)- 8.7<TE (kPa) <14.4; Historical control-Corpechot et al 2014
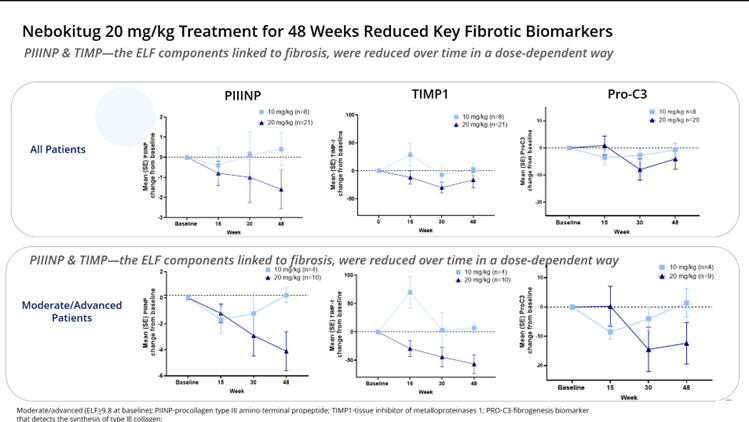
Moderate/advanced (ELF>9.8 at baseline); PIINP-procollagen type III amino-terminal propeptide; TIMP1-tissue inhibitor of metalloproteinases 1; PRO-C3-fibrogenesis biomarker that detects the synthesis of type lll collagen
Dr. Mor continued, “We view the broad and consistent stabilization and improvement in disease-related biomarkers seen with 48 weeks of nebokitug treatment, including reductions in the risk of disease progression as shown by the ELF score, as directly translatable and potentially derisking to our Phase 3 study. Additionally, the new data showed a significantly lower rate of clinical events in patients treated with nebokitug for 48 weeks compared to historical controls, further increasing our confidence in the Phase 3 trial design, where the primary endpoint is based on the number of clinical events. These findings reinforce nebokitug’s potential to alter the course of PSC, as well as the rationale for the planned Phase 3 trial that provides a clear pathway to potential full regulatory approval.”
Douglas Thorburn, MD, Divisional Clinical Director for Liver and Digestive Health at the Royal Free London NHS Trust and Professor of Hepatology within the Institute for Liver and Digestive Health at UCL, and a coordinating investigator of the SPRING trial, said, “This new data reporting on patients who received up to 48 weeks of treatment with nebokitug reinforces the positive results that we saw in the initial 15-week double blinded trial. The fact that nebokitug continued to be generally well tolerated and to generate continued improvements across a broad range of disease-related biomarkers is very encouraging. Patients with PSC experience burdensome symptoms, unpredictable futures and lack effective treatments. I look forward to the continued clinical progress of nebokitug as a potential new therapy for PSC where there is so much unmet medical need.”
Further details on the SPRING Trial Open Label Extension results can be viewed at https://investors.chemomab.com/presentations
About the Nebokitug SPRING Trial Open Label Extension (OLE)
The SPRING trial OLE was available to PSC patients who enrolled in the nebokitug Phase 2 SPRING trial (NCT04595825) after the OLE was added to the study design, and who completed the 15-week double-blind placebo-controlled portion of the trial. Of those 54 patients eligible to roll over,
About Nebokitug (CM-101)
Nebokitug is a first-in-class dual activity monoclonal antibody that neutralizes CCL24, a soluble protein that helps drive the inflammatory and fibrotic pathways central to primary sclerosing cholangitis (PSC) and other fibro-inflammatory diseases. By inhibiting CCL24, nebokitug blocks immune cell recruitment and fibroblast activation, thereby interrupting the self-reinforcing cycle that results in fibrosis. In clinical and preclinical studies, nebokitug has been shown to have a favorable safety profile, with the potential to treat multiple severe and life-threatening fibro-inflammatory diseases. Chemomab has reported positive results from four clinical trials of nebokitug in patients, including the Phase 2 SPRING trial in patients with PSC. This study achieved the primary safety endpoint and nebokitug-treated patients with moderate/advanced disease showed improvements on a wide range of disease-related secondary endpoint. The results of the Open Label Extension portion of the SPRING trial, where PSC patients received nebokitug for up to a total of 48 weeks, confirmed and extended the initial Phase 2 results. Nebokitug is also potentially being developed for systemic sclerosis (SSc). The SSc program has an open U.S. IND. Nebokitug has received FDA and EMA Orphan Drug designations for the treatment of PSC and SSc and FDA Fast Track status for the treatment of PSC in adults.
About Primary Sclerosing Cholangitis
PSC is a rare, debilitating progressive liver disease characterized by inflammation and fibrosis (scarring) of the bile ducts that can lead to cirrhosis of the liver, liver failure and death. PSC also increases the risk of various cancers, which account for about half of PSC-related mortality. PSC affects an estimated 30,000 patients in the U.S. and about 80,000 worldwide. The underlying cause of PSC is unknown, but about
1 - Muir et al, 2019
Forward-Looking Statements
This press release contains forward-looking statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995 that involve substantial risks and uncertainties. All statements other than statements of historical facts contained in this press release, including statements regarding our future financial condition, results of operations, business strategy and plans, and objectives of management for future operations, as well as statements regarding industry trends, are forward-looking statements. In some cases, you can identify forward-looking statements by terminology such as “estimate,” “intend,” “may,” “plan,” “potentially” “will” or the negative of these terms or other similar expressions. We have based these forward-looking statements largely on our current expectations and projections about future events and trends that we believe may affect our financial condition, results of operations, business strategy and financial needs. These forward-looking statements are subject to a number of risks, uncertainties and assumptions, including, among other things: the risk that certain acknowledgements from the End-of-Phase 2 (EOP2) meeting with the FDA in connection with PSC regulatory approval will not materialize into a pathway for regulatory approval; that certain conclusions and assumptions drawn from the EOP2 meeting with the FDA discussed in the presentation will prove incorrect and adversely affect the ability for nebokitug to become an FDA fully approved therapy; the risk that the full data set from the nebokitug study or data generated in further clinical trials of nebokitug will not be consistent with the topline results of the nebokitug Phase 2 PSC trial; failure to obtain, or delays in obtaining, regulatory approvals for nebokitug in the U.S., Europe or other territories; failure to successfully commercialize nebokitug, if approved by applicable regulatory authorities, in the U.S., Europe or other territories, or to maintain U.S., European or other territory regulatory approval for nebokitug if approved; uncertainties in the degree of market acceptance of nebokitug by physicians, patients, third-party payors and others in the healthcare community; nebokitug development of unexpected safety or efficacy concerns related to nebokitug; failure to successfully conduct future clinical trials for nebokitug, including due to the Company's potential inability to enroll or retain sufficient patients to conduct and complete the trials or generate data necessary for regulatory approval, among other things; risks that the Company's clinical studies will be delayed or that serious side effects will be identified during drug development; failure of third parties on which the Company is dependent to manufacture sufficient quantities of nebokitug for commercial or clinical needs, to conduct the Company's clinical trials; changes in laws and regulations applicable to the Company's business and failure to comply with such laws and regulations; business or economic disruptions due to catastrophes or other events, including natural disasters or public health crises; and uncertainties with respect to the Company's need and ability to access future capital; and the intensity and duration of the current war in Israel, and its impact on our operations in Israel. These risks are not exhaustive. You should carefully consider the risks and uncertainties described in the “Risk Factors” sections of our 20-F for the year ended December 31, 2024. New risk factors emerge from time to time, and it is not possible for our management to predict all risk factors, nor can we assess the impact of all factors on our business or the extent to which any factor, or combination of factors, may cause actual results to differ materially from those contained in, or implied by, any forward-looking statements. You should not rely upon forward-looking statements as predictions of future events. Except as required by law, we undertake no obligation to update publicly any forward-looking statements for any reason after the date of this press release. Before you invest, you should read the documents we have filed and will file with the SEC for more complete information about us. You may get these documents for free by visiting EDGAR on the SEC website at www.sec.gov. This press release shall not constitute an offer to sell or the solicitation of an offer to buy these securities, nor shall there be any sale of these securities in any state or jurisdiction in which such offer, solicitation, or sale would be unlawful prior to registration or qualification under the securities law of any such state or jurisdiction.
About Chemomab Therapeutics Ltd.
Chemomab is a clinical stage biotechnology company developing innovative therapeutics for fibro-inflammatory diseases with high unmet need. Based on the unique role of the soluble protein CCL24 in promoting fibrosis and inflammation, Chemomab developed nebokitug (CM-101), a first-in-class dual activity monoclonal antibody that neutralizes CCL24 and has demonstrated disease-modifying potential. In clinical and preclinical studies, nebokitug has been shown to have a favorable safety profile and has been generally well-tolerated, with the potential to treat multiple severe and life-threatening fibro-inflammatory diseases. Chemomab has reported positive results from four clinical trials of nebokitug in patients. Based on positive data from its Phase 2 SPRING trial in primary sclerosing cholangitis (PSC), the company is preparing for potential initiation of a nebokitug PSC Phase 3 trial. The design of Phase 3 calls for a single pivotal trial based on a clinical event primary endpoint that provides a clear and streamlined pathway to potential full regulatory approval. Nebokitug has received FDA and EMA Orphan Drug and FDA Fast Track designations for the treatment of PSC. Chemomab’s nebokitug program for the treatment of systemic sclerosis has an open U.S. IND. For more information, visit: chemomab.com.
Contacts:
Media and Investors:
Barbara Lindheim
Consulting Vice President, Investor & Public Relations, Strategic Communications
Phone: +1 917-355-9234
barbara.lindheim@chemomab.com
IR@chemomab.com
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/1e169756-83fa-4933-b717-0b778ab8c3c2
https://www.globenewswire.com/NewsRoom/AttachmentNg/b03c0e26-84e7-4363-8d6e-4865acdb0678
https://www.globenewswire.com/NewsRoom/AttachmentNg/f1e1f110-80ee-4495-bb1c-c6fcdc1e234c








