Aldeyra Therapeutics Announces Advancement of New RASP Modulators and Recent Preclinical Data in Obesity at 2024 Investor Roundtable
Aldeyra Therapeutics announced significant advancements in its RASP modulator pipeline and recent preclinical obesity data at its 2024 Investor Roundtable. Key updates include ADX-629 moving to pediatric Phase 2 trials for Sjögren-Larsson Syndrome, with top-line results expected in 2025. ADX-743, showing promising results in lowering triglycerides and fatty acids, is advancing to IND-enabling studies, with an application expected in 2025. ADX-248 will start Phase 1 clinical testing for atopic dermatitis in the second half of 2024. ADX-631 begins preclinical testing for retinal diseases, with an IND application planned for 2025. CEO Todd C. Brady emphasized the expansion of their RASP modulator pipeline for treating inflammatory and metabolic diseases. The Investor Roundtable, including a live webcast, is scheduled for today at 8:00 a.m. ET, with a replay available for 90 days.
- Advancement of ADX-629 to pediatric Phase 2 trials for Sjögren-Larsson Syndrome, with top-line results expected in 2025.
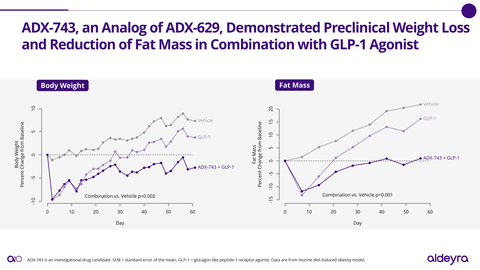
- Promising preclinical obesity data leading to ADX-743 advancement to IND-enabling studies, with an application expected in 2025.
- ADX-248 advancing to Phase 1 clinical testing for atopic dermatitis in the second half of 2024.
- Initiation of preclinical testing for ADX-631 in retinal diseases, with an IND application planned for the first half of 2025.
- Top-line results for ADX-629 in pediatric trials for Sjögren-Larsson Syndrome won't be available until 2025, indicating a long development timeline.
- Potential delays or adverse outcomes in clinical trials could impact the company's progress and investor confidence.
Insights
Aldeyra's advancements with their new RASP modulators signify important progress in addressing complex inflammatory and metabolic diseases. The initiation of ADX-629 into the pediatric cohort for Sjögren-Larsson Syndrome is noteworthy. This rare genetic condition has limited treatment options and seeing near-normalization of lipid profiles in adults is promising. These advancements are early stage, but if results in children mirror those in adults, it could mark a significant breakthrough.
Additionally, the preclinical data for ADX-743 in obesity and its advancement towards IND-enabling studies highlights Aldeyra's proactive strategy in tackling common metabolic disorders. Obesity and hypertriglyceridemia are widespread issues and innovative treatments are necessary. However, it is important to observe that these remain in preclinical stages and translating these results to clinical success is not guaranteed.
Finally, ADX-248's progression to Phase 1/2 clinical trials for atopic dermatitis, based on its superior preclinical results, is encouraging. Atopic dermatitis affects a vast number of individuals and having more effective treatment options could greatly benefit patients.
From a financial perspective, Aldeyra Therapeutics demonstrates a robust and ambitious pipeline. Progressing RASP modulators into various stages of clinical and preclinical trials showcases a diversified strategy. This diversified approach can attract investors due to the potential for multiple revenue streams if these treatments prove successful.
However, investors should be cautious. Clinical trials, especially in early stages, are high-risk with substantial cost implications. The anticipated top-line results for ADX-629 in pediatric patients by 2025 and the potential IND applications for ADX-743 and ADX-631 by the same year indicate a timeline that requires patience. Near-term financial returns may not be immediate and continued investment in R&D will be necessary.
Despite these risks, positive results could lead to substantial market opportunities, especially considering the size of the markets for obesity, atopic dermatitis and retinal diseases.
Live Webcast Scheduled to Begin at 8:00 a.m. ET Today
(Graphic: Aldeyra)
Pipeline Updates
- Following positive biomarker results in adults, including near-normalization of lipid profiles, ADX-629, a first-in-class orally administered investigational RASP modulator, advanced to the pediatric cohort of the Phase 2 clinical trial in Sjögren-Larsson Syndrome, a rare inborn error of metabolism caused by mutations in fatty aldehyde dehydrogenase and characterized by cognitive dysfunction and skin disorders. Top-line results from approximately five pediatric patients are expected in 2025.
- Based on new results in a preclinical model of obesity, novel RASP modulator ADX-743 was advanced to Investigational New Drug (IND)-enabling studies. ADX-743 is an analog of ADX-629, which has demonstrated lowering of triglycerides and fatty acids in Phase 1 and Phase 2 clinical trials. Pending additional results, Aldeyra expects to submit an IND application for ADX-743 or an alternative RASP modulator for obesity or hypertriglyceridemia in 2025.
- Based in part on superior preclinical results in atopic dermatitis and unprecedented exposure in preclinical RASP modulator pharmacokinetic models, ADX-248 will be advanced in lieu of ADX-246 to Phase 1/2 clinical testing in atopic dermatitis. Phase 1 clinical testing of ADX-248 is expected to begin in the second half of 2024.
- Novel RASP modulator ADX-631 has initiated preclinical testing in models of retinal disease. ADX-631 is an analog of ADX-103, which previously demonstrated activity in animal models of the dry form of age-related macular degeneration (dry AMD), retinal inflammation, and diabetic macular edema. Aldeyra expects to submit an IND application for ADX-631 or an alternative RASP modulator for the treatment of dry AMD or geographic atrophy in the first half of 2025.
“Consistent with the new data presented at our Research and Development Day in April of this year, we continue to expand our novel RASP modulator pipeline with the discovery and advancement of new RASP modulators for the treatment of inflammatory and metabolic diseases,” stated Todd C. Brady, M.D., Ph.D., President and CEO of Aldeyra. “We look forward to discussing our recent progress on today’s call with investors and are excited to share subsequent updates on our pipeline throughout the remainder of 2024.”
Investor Roundtable Webcast Information
The 2024 Aldeyra Therapeutics Investor Roundtable will take place at 8:00 a.m. ET today, Thursday, June 20, 2024. A live audio webcast will be accessible from the “Investors & Media” section of the Aldeyra website at https://ir.aldeyra.com/. A replay will be available for 90 days following the event.
About Aldeyra
Aldeyra Therapeutics is a biotechnology company devoted to discovering innovative therapies designed to treat immune-mediated and metabolic diseases. Our approach is to develop pharmaceuticals that modulate protein systems, instead of directly inhibiting or activating single protein targets, with the goal of optimizing multiple pathways at once while minimizing toxicity. Our product candidates include RASP (reactive aldehyde species) modulators ADX-629, ADX-248, ADX-743, ADX-631, and chemically related molecules for the potential treatment of systemic and retinal immune-mediated and metabolic diseases. Our late-stage product candidates are reproxalap, a RASP modulator for the potential treatment of dry eye disease and allergic conjunctivitis, and ADX-2191, a novel formulation of intravitreal methotrexate for the potential treatment of retinitis pigmentosa.
Safe Harbor Statement
This release contains forward-looking statements within the meaning of the Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding Aldeyra’s future expectations, plans, and prospects, including without limitation statements regarding: the goals, opportunity, and potential for Aldeyra’s product candidates; the outcome and expected timing and the results of Aldeyra’s planned preclinical and clinical trials, including planned and ongoing trials; the outcome and timing of the FDA’s review, acceptance and/or approval of IND submissions for Aldeyra’s product candidates. Aldeyra intends such forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 21E of the Securities Exchange Act of 1934 and the Private Securities Litigation Reform Act of 1995. In some cases, you can identify forward-looking statements by terms such as, but not limited to, “may,” “might,” “will,” “objective,” “intend,” “should,” "could," “can,” “would,” “expect,” “believe,” “anticipate,” “project,” “on track,” “scheduled,” “target,” “design,” “estimate,” “predict,” “contemplates,” “likely,” “potential,” “continue,” “ongoing,” “aim,” “plan,” or the negative of these terms, and similar expressions intended to identify forward-looking statements. Such forward-looking statements are based upon current expectations that involve risks, changes in circumstances, assumptions, and uncertainties. Aldeyra is at an early stage of development and may not ever have any products that generate significant revenue. All of Aldeyra's development timelines may be subject to adjustment depending on recruitment rate, regulatory review, preclinical and clinical results, funding, and other factors that could delay the initiation, enrollment, or completion of clinical trials. Important factors that could cause actual results to differ materially from those reflected in Aldeyra's forward-looking statements include, among others, the timing of enrollment, commencement and completion of Aldeyra's clinical trials, the timing and success of preclinical studies and clinical trials conducted by Aldeyra and its development partners; delay in or failure to obtain regulatory approval of Aldeyra's product candidates, including as a result of the FDA not accepting Aldeyra’s regulatory filings, issuing a complete response letter, or requiring additional clinical trials or data prior to review or approval of such filings or in connection with resubmissions of such filings; the ability to maintain regulatory approval of Aldeyra's product candidates, and the labeling for any approved products; the risk that prior results, such as signals of safety, activity, or durability of effect, observed from preclinical or clinical trials, will not be replicated or will not continue in ongoing or future studies or clinical trials involving Aldeyra's product candidates in clinical trials focused on the same or different indications; the scope, progress, expansion, and costs of developing and commercializing Aldeyra's product candidates; uncertainty as to Aldeyra’s ability to commercialize (alone or with others) and obtain reimbursement for Aldeyra's product candidates following regulatory approval, if any; the size and growth of the potential markets and pricing for Aldeyra's product candidates and the ability to serve those markets; Aldeyra's expectations regarding Aldeyra's expenses and future revenue, the timing of future revenue, the sufficiency or use of Aldeyra's cash resources and needs for additional financing; the rate and degree of market acceptance of any of Aldeyra's product candidates; Aldeyra's expectations regarding competition; Aldeyra's anticipated growth strategies; Aldeyra's ability to attract or retain key personnel; Aldeyra’s commercialization, marketing and manufacturing capabilities and strategy; Aldeyra's ability to establish and maintain development partnerships; Aldeyra’s ability to successfully integrate acquisitions into its business; Aldeyra's expectations regarding federal, state, and foreign regulatory requirements; political, economic, legal, social, and health risks, public health measures, and war or other military actions, that may affect Aldeyra’s business or the global economy; regulatory developments in
In addition to the risks described above and in Aldeyra's other filings with the SEC, other unknown or unpredictable factors also could affect Aldeyra's results. No forward-looking statements can be guaranteed and actual results may differ materially from such statements. The information in this release is provided only as of the date of this release, and Aldeyra undertakes no obligation to update any forward-looking statements contained in this release on account of new information, future events, or otherwise, except as required by law.
View source version on businesswire.com: https://www.businesswire.com/news/home/20240620180926/en/
Investor & Media Contact:
David Burke
Tel: (917) 618-2651
investorrelations@aldeyra.com
Source: Aldeyra Therapeutics, Inc.







